BITSAT 2009 Question Paper PDF is available for download. BITSAT 2009 was conducted in online CBT mode by BITS Pilani. BITSAT 2009 Question Paper had 150 questions to be attempted in 3 hours.
BITSAT 2009 Question Paper with Answer Key PDF
| BITSAT 2009 Question Paper with Solution PDF | Download PDF | Check Solutions |
Given that \( \vec{A} + \vec{B} = \vec{R} \) and \( A^2 + B^2 = R^2 \). The angle between \( \vec{A} \) and \( \vec{B} \) is:
In the relation \( P = \frac{\alpha Z}{\beta} e^{-k\theta} \), \(P\) is pressure, \(Z\) is distance, \(k\) is Boltzmann constant and \(\theta\) is the temperature. The dimensional formula of \(\beta\) will be:
Which of the following is most accurate?
A projectile projected at an angle \(30^\circ\) from the horizontal has a range \(R\). If the angle of projection at the same initial velocity is \(60^\circ\), the range will be:
A block of mass \(M\) is pulled along a horizontal frictionless surface by a rope of mass \(M/2\). If a force \(2Mg\) is applied at one end of the rope, the force which the rope exerts on the block is:
A chain of mass \(M\) is placed on a smooth table with \(1/n\) of its length hanging over the edge. The work done in pulling the hanging portion of the chain back to the surface of the table is:
A particle of mass 10 kg moving eastwards with a speed 5 m s\(^{-1}\) collides with another particle of same mass moving northwards with the same speed. The two particles coalesce on collision. The new particle of mass 20 kg will move in the north-east direction with velocity:
A uniform cube of side \(a\) and mass \(m\) rests on a rough horizontal table. A horizontal force \(F\) is applied normal to one of the faces at a point directly above the centre of the face, at a height \(3a/4\) above the base. The minimum value of \(F\) for which the cube begins to topple on an edge is (assume the cube does not slide):

The rotation of the earth having radius \(R\) about its axis speeds upto a value such that a man at latitude angle \(60^\circ\) feels weightless. The duration of the day in such case will be:
A metallic rod breaks when strain produced is \(0.2%\). The Young’s modulus of the material of the rod is \(7\times10^9\ N m^{-2}\). What should be its area of cross-section to support a load of \(10^4\ N\)?
A liquid is flowing through a non-sectional tube with its axis horizontal. If two points X and Y on the axis of tube have sectional area 2.0 cm\(^2\) and 25 mm\(^2\) respectively then find the flow velocity at Y when the flow velocity at X is 10 m/s.
A body of length 1 m having cross-sectional area 0.75 m\(^2\) has heat flow through it at the rate of 600 Joule/sec. Find the temperature difference if \(K = 200\ J m^{-1}K^{-1}\).
Which of the following combinations of properties would be most desirable for a cooking pot?
A particle starts moving rectilinearly at time \(t=0\) such that its velocity \(v\) changes with time \(t\) according to the equation \(v=t^2-t\), where \(t\) is in seconds and \(v\) is in m/s. Find the time interval for which the particle retards.
A sample of gas expands from volume \(V_1\) to \(V_2\). The amount of work done by the gas is greatest when the expansion is:
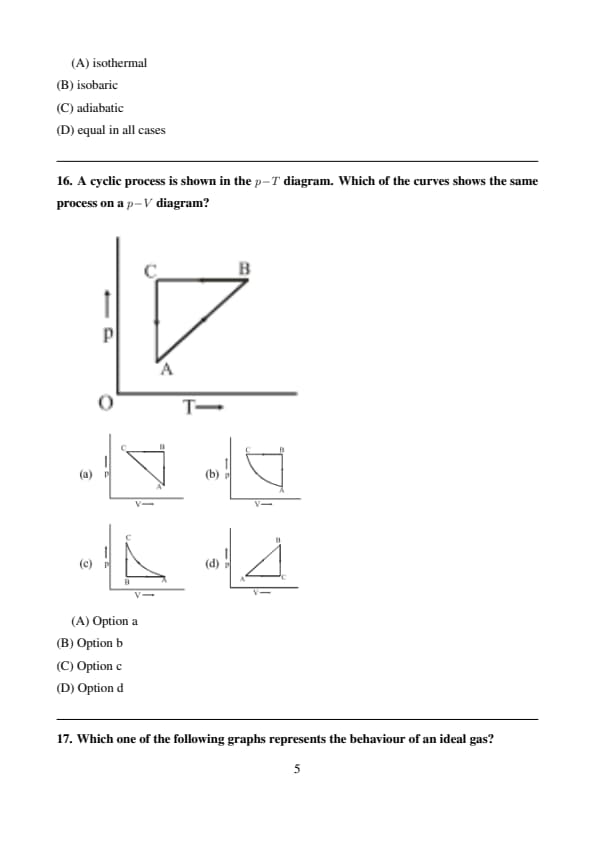
A cyclic process is shown in the \(p\!-\!T\) diagram. Which of the curves shows the same process on a \(p\!-\!V\) diagram?


Which one of the following graphs represents the behaviour of an ideal gas?

In case of a forced vibration, the resonance curve becomes very sharp when the
A pendulum bob carries a +ve charge \(+q\). A positive charge \(+q\) is held at the point of support. Then the time period of the bob is (where \(L\) = length of pendulum, \(g_{eff}\) = effective value of \(g\)):
Two tuning forks A and B sounded together give 6 beats per second. When at resonance tube closed at one end, the two forks give resonance when the air columns are 24 cm and 25 cm respectively. Calculate the frequencies of forks.
An electron has an initial velocity in a direction different from that of an electric field, the path of the electron is
If on combining two charged bodies, the current does not flow then
Calculate the area of the plates of a one farad parallel plate capacitor if separation between plates is 1 mm and plates are in vacuum
The length of a potentiometer wire is \(\ell\). A cell of emf \(E\) is balanced at a length \(\ell/3\) from the positive end of the wire. If the length of the wire is increased by \(\ell/2\). At what distance will the same cell give a balance point?
A constant current \(i\) flows in a loop of radius \(r\). It is placed in a uniform magnetic field \(\vec{B}_0\) such that \(\vec{B}_0\) is perpendicular to the plane of the loop. The magnetic force acting on the loop is
An ammeter reads upto 1 ampere. Its internal resistance is 0.81 ohm. To increase the range to 10 A the value of required shunt is
At the magnetic north pole of the earth, the value of horizontal component of earth’s magnetic field and angle of dip are, respectively
Lenz’s law is a consequence of the law of conservation of
The instantaneous current from an a.c. source is \(i = 6\sin314t\). What is the rms value of the current?
A coil has resistance 30 ohm and inductive reactance 20 ohm at 50 Hz frequency. If an a.c. source of 200 volt, 100 Hz is connected across the coil, the current in the coil will be
The magnetic field in a travelling electromagnetic wave has a peak value of 20 nT. The peak value of electric field strength is
A plano-convex lens of focal length 30 cm has its plane surface silvered. An object is placed 40 cm from the lens on the convex side. The distance of the image from the lens is
When a mica sheet of thickness 7 microns and \(\mu=1.6\) is placed in the path of one of interfering beams in a biprism experiment then the central fringe gets at the position of seventh bright fringe. The wavelength of light used will be
In Young's double slit experiment, if the slit widths are in the ratio 1:2, the ratio of the intensities at maxima will be
In a photoelectric experiment, with light of wavelength \(\lambda\), the fastest electron has speed \(v\). If the exciting wavelength is changed to \(3\lambda/4\), the speed of the fastest emitted electron will become
Taking Rydberg’s constant \(R_H=1.097\times10^7\ m^{-1}\), the first and second wavelength of Balmer series in hydrogen spectrum is
An X-ray tube is operated at 15 kV. Calculate the upper limit of the speed of the electrons striking the target.
Nuclear energy released in fission since binding energy per nucleon is
Assuming the diodes to be of silicon with forward resistance zero, the current \(I\) in the following circuit is

The truth table given below corresponds to the logic gate
\[ \begin{array}{c|c|c} A & B & X
\hline 0 & 0 & 1
1 & 0 & 0
0 & 1 & 0
1 & 1 & 0 \end{array} \]
Given the numbers : 161 cm, 0.161 cm, 0.0161 cm. The number of significant figures for the three numbers are
Beryllium resembles much with :
Which one of the following ions has the highest value of ionic radius?
Which of the following two are isostructural ?
The cooking in a pressure cooker is less because :
For the reaction : \( \mathrm{N_2 + 3H_2 \rightleftharpoons 2NH_3} \). Which one of the following is correct regarding \(\Delta H\):
One mole of an ideal gas at 300 K is expanded isothermally from an initial volume of 1 litre to 10 litres. The \(\Delta E\) for this process is (R = 2 cal mol\(^{-1}\) K\(^{-1}\)):
At 25°C and 1 bar which of the following has a non-zero \(\Delta H_f^\circ\) ?
If the equilibrium constant of the reaction \(2HI \rightleftharpoons H_2 + I_2\) is 0.25, then the equilibrium constant for the reaction \(H_2 + I_2 \rightleftharpoons 2HI\) would be
The oxidation states of sulphur in the anions \(\mathrm{SO_3^{2-}},\ \mathrm{S_2O_4^{2-}}\) and \(\mathrm{S_2O_6^{2-}}\) follow the order
The value of x is maximum for
For making good quality mirrors, plates of float glass are used. These are obtained by floating molten glass over a liquid metal which does not solidify before glass. The metal used can be
The intermediate formed during the addition of HCl to propene in the presence of peroxide is
Which of the following has zero dipole moment?
Keto–enol tautomerism is observed in

Which one of the following contain isopropyl group?
The statement which is not correct about control of particulate pollution is:
Chief source of soil and water pollution is:
The false statement among the followings:
The atomic radius of atom is \(r\). Total volume of atoms present in an fcc unit cell of an element is:
Which one of the following statements is false?
The degree of dissociation of Ca(NO\(_3\))\(_2\) in a dilute aqueous solution containing 7.0 g of salt per 100 g of water at \(100^\circ\)C is 70%. If vapour pressure of water at \(100^\circ\)C is 760 mm Hg, the vapour pressure of the solution is:
When the sample of copper with zinc impurity is to be purified by electrolysis, the appropriate electrodes are:
\begin{tabular{c c
Cathode & Anode
\end{tabular
The conductivity of a saturated solution of BaSO\(_4\) is \(3.06 \times 10^{-6}\ \Omega^{-1}cm^{-1}\) and its equivalent conductance is \(1.53\ \Omega^{-1}cm^2eq^{-1}\). The \(K_{sp}\) for BaSO\(_4\) is:
A cell that utilises the reaction \[ Zn(s) + 2H^+(aq) \rightleftharpoons Zn^{2+}(aq) + H_2(g) \]
addition of H\(_2\)SO\(_4\) to cathode compartment, will:
The chemical reaction \(2O_3 \rightarrow 3O_2\) proceeds as follows: \[ O_3 \xrightarrow{fast} O_2 + O \] \[ O + O_3 \xrightarrow{slow} 2O_2 \]
The rate law expression should be:
Among the following statements the incorrect one is:
Cinnabar is an ore of:
Which of the following is used in the preparation of chlorine?
Which of the following elements does not belong to the first transition series?
[EDTA]\(^{4-}\) is a:
Which of the following order is not correct?
When esters are hydrolysed the product which gives hydrogen ions is:
Which of the following compound cannot be used in preparation of iodoform?
Which of the following compound is obtained by heating ammonium cyanate?
Which of the following statements about vitamin B\(_{12}\) is incorrect?
Ammonia forms the complex ion [Cu(NH\(_3\))\(_4\)]\(^{2+}\) with copper ions in alkaline solutions but not in acidic solutions. What is the reason for it?
An aqueous solution of a substance gives a white precipitate on treatment with dil. HCl which dissolves on heating. When H\(_2\)S is passed through the hot acidic solution, a black precipitate is obtained. The substance is a:
The one which is least basic is:
Interparticle forces present in nylon-66 are:
If \(A=\{1,2,3,4,5\}\), then the number of proper subsets of \(A\) is:
The range of the function \(f(x)=\dfrac{x^2-x+1}{x^2+x+1}\), where \(x\in\mathbb{R}\), is:
If \(y=\dfrac{2\sin\alpha}{1+\cos\alpha+\sin\alpha}\), then the value of \(\dfrac{1-\cos\alpha+\sin\alpha}{1+\sin\alpha}\) is:
The period of \(\dfrac{\sin\theta+\sin2\theta}{\cos\theta+\cos2\theta}\) is:
The general solution of \(8\tan^2\frac{x}{2}=1+\sec x\) is:
\(10^n+3(4^{n+2})+5\) is divisible by (\(n\in\mathbb{N}\)):
If the expression \(x^2-11x+a\) and \(ax^2-14x+2a\) must have a common factor, then the common factor is:
For the equation \(\dfrac{1}{x+a}+\dfrac{1}{x+b}=\dfrac{1}{x+c}\), if the product of roots is zero, the sum of roots is:
If \(\arg(z_1)=\arg(z_2)\), then:
If \(\dfrac{2x+3}{5} < \dfrac{4x-1}{2}\), then \(x\) lies in the interval:
The letters of the word TOUGH are written in all possible orders and the words are written out as in a dictionary. Then the rank of the word TOUGH is:
If in the expansion of \(\left(2x+\dfrac{1}{4x}\right)^n\), \(T_3 = 7T_2\) and the sum of the binomial coefficients of second and third terms is 36, then the value of \(x\) is:
The 100th term of the sequence \(1,2,2,3,3,3,4,4,4,4,\dots\) is:
View Solution
The number \(n\) appears \(n\) times.
Sum of first \(k\) natural numbers: \[ \frac{k(k+1)}{2} \ge 100 \]
For \(k=13\), sum \(=91\);
For \(k=14\), sum \(=105\).
Hence the 100th term is 14. Quick Tip: Use triangular numbers to locate terms.
The line \(3x-4y+7=0\) is rotated through an angle \(\frac{\pi}{4}\) in the clockwise direction about the point \((-1,1)\). The equation of the line in its new position is:
Find the vertex of the parabola \(x^2-8y-x+19=0\).
If \(f(t)=\dfrac{1-t}{1+t}\), then \(f'(1)\) is equal to:
If: p: Raju is tall and q: Raju is intelligent, then the symbolic statement \(\sim p \vee q\) means:
Given below is a frequency distribution with median 46. In this distribution, some of the frequencies are missing. Determine the missing frequencies.
\begin{tabular{|c|c|c|c|c|c|c|c|
\hline
Marks & 10--20 & 20--30 & 30--40 & 40--50 & 50--60 & 60--70 & 70--80
\hline
No. of students & 12 & 30 & \(x\) & 65 & \(y\) & 25 & 18
\hline
\end{tabular
Total number of students = 229
If the function \(f:(-\infty,\infty)\to B\) defined by \(f(x)=-x^2+6x-8\) is bijective, then \(B=\)
Find the value of \[ 2\tan^{-1}\frac{1}{5}+\tan^{-1}\frac{1}{2}+2\tan^{-1}\frac{1}{8} \]
If \(A\) and \(B\) are \(2\times2\) matrices, which of the following is true?
If \(a>0,\ b>0,\ c>0\) are respectively the \(p\)th, \(q\)th, \(r\)th terms of G.P., then the value of the determinant \[ \begin{vmatrix} \log a & p & 1
\log b & q & 1
\log c & r & 1 \end{vmatrix} \]
is
The digits \(A,B,C\) are such that the three-digit numbers \(A88, 6B8, 86C\) are divisible by 72. Then the determinant \[ \begin{vmatrix} A & 6 & 8
8 & B & 6
8 & 8 & C \end{vmatrix} \]
is divisible by
If \[ M(\alpha)= \begin{bmatrix} \cos\alpha & -\sin\alpha & 0
\sin\alpha & \cos\alpha & 0
0 & 0 & 1 \end{bmatrix}, \quad M(\beta)= \begin{bmatrix} \cos\beta & 0 & \sin\beta
0 & 1 & 0
-\sin\beta & 0 & \cos\beta \end{bmatrix} \]
then \([M(\alpha)M(\beta)]^{-1}\) is equal to
If \(y=e^{-x}\cos x\) and \(y+k y'=0\), where \(y_4=\dfrac{d^4y}{dx^4}\), then \(k=\)
The set of points of discontinuity of the function \[ f(x)=\frac{1}{|x|\log|x|} \]
is
The minimum value of the function \(y=x^4-2x^2+1\) in the interval \(\left[-\frac12,2\right]\) is
The value of \(a\) in order that \[ f(x)=\sin x-\cos x-ax+b \]
decreases for all real values is
The equation of tangent to the curve \(y=\sin x\) at the point \((\pi,0)\) is
If \[ \int \frac{2\cos x-\sin x+\lambda}{\cos x+\sin x-2}\,dx = A\ln|\cos x+\sin x-2|+Bx+C, \]
then the ordered triplet \((A,B,\lambda)\) is
Evaluate: \[ \int x\tan^{-1}x\,dx \]
Evaluate \[ \int_0^1 \frac{dx}{\sqrt{2-x^2}} \]
If \(\int_0^n [x]\,dx = 66\), then \(n=\)
Area of the triangle formed by the line \(x+y=3\) and angle bisectors of the pair of straight lines \(x^2-y^2+2y=1\) is
Solution of differential equation \[ \frac{dy}{dx}+\frac{y}{x}=\sin x \]
If the line \[ \frac{x-4}{1}=\frac{y-2}{1}=\frac{z-k}{2} \]
lies in the plane \(2x-4y+z=7\), then the value of \(k\) is
A line segment has length 63 and direction ratios are \(3,-2,6\). If the line makes an obtuse angle with x-axis, the components of the line vector are
It is given that the events \(A\) and \(B\) are such that \[ P(A)=\frac14,\ P(A|B)=\frac12,\ P(B|A)=\frac23 \]
Then \(P(B)\) is
The random variable \(X\) has the following probability distribution
\begin{tabular{|c|c|c|c|c|c|
\hline \(x\) & 0 & 1 & 2 & 3 & 4
\hline \(P(X=x)\) & \(k\) & \(3k\) & \(5k\) & \(2k\) & \(k\)
\hline
\end{tabular
Then the value of \(P(X\ge2)\) is
In a triangle \(ABC\), \(\angle C=90^\circ\), then \[ \frac{a^2-b^2}{a^2+b^2} \]
is equal to
A person standing on the bank of a river observes that the angle subtended by a tree on the opposite bank is \(60^\circ\). When he retreats 20 feet from the bank, he finds the angle to be \(30^\circ\). The breadth of the river in feet is:
The minimum value of the function \(z=4x+3y\) subject to the constraints \[ 3x+2y\ge160,\;5x+2y\ge200,\;x+2y\ge80,\;x\ge0,\;y\ge0 \]
is
If \(|r|>1\) and \[ x=a+\frac{a}{r^2}+\frac{a}{r^4}+\cdots,\quad y=b-\frac{b}{r^2}+\frac{b}{r^4}-\cdots, \] \[ z=c+\frac{c}{r^2}+\frac{c}{r^4}+\cdots, \]
then \(\dfrac{xy}{z}\) is equal to
Two tangents \(PQ\) and \(PR\) drawn to the circle \(x^2+y^2-2x-4y-20=0\) from point \(P(16,7)\). If the centre of the circle is \(C\), the area of quadrilateral \(PQCR\) is
The value of \[ \lim_{x\to0}\frac{(4^x-1)^3}{x^2\log(1+3x)} \]
is
Agnostic
Bohemian
Cacographist
Spelling test – find correct spelling:
Spelling test – find correct spelling:
Spelling test – find correct spelling:
REPRIMAND
IMPERTINENT
EQUIVOCAL
It is difficult to believe what he tells us because his account of any event is always full of _________.
The bank clerk tried to _________ money from his friend’s account.
Eight scientists have _________ the national awards for outstanding contribution and dedication to the profession.
Freedom, is the restricted kind in the sense (P), the rich and poor woman (Q), that a wide gulf separates (R), which a modern woman enjoys (S).
In life, some rules are (P), as in business (Q), they seem almost instinctive (R), learnt so early that (S).
Kapil, left in an aeroplane (P), after reading a sailing magazine (Q), had decided (R), to build his own boat nine years earlier (S).
Distance : Odometer :: ? : Barometer
One of the numbers does not fit into the series. Find the wrong number. \[ 13,\;16,\;38,\;124,\;504,\;2535 \]
Statement:
In order to reduce the gap between income and expenditure, the company has decided to increase the price of its product from next month.
Assumptions:
I. The rate will remain more or less same after the increase.
II. The expenditure will more or less remain the same in near future.
III. The rival companies will also increase the price of the similar product.
FLMO : ?? :: BFEN : ARSO
If A denotes '+', B denotes '-', C denotes '×'. Then what is the value of \[ 10\ C\ 4\ A\ (4\ C\ 4)\ B\ 6? \]
In this question, two figures are given to the left of the sign :: and one figure is given to the right of the sign. Find the correct alternative.

Identify the missing part of the figure and select it from the given alternatives.

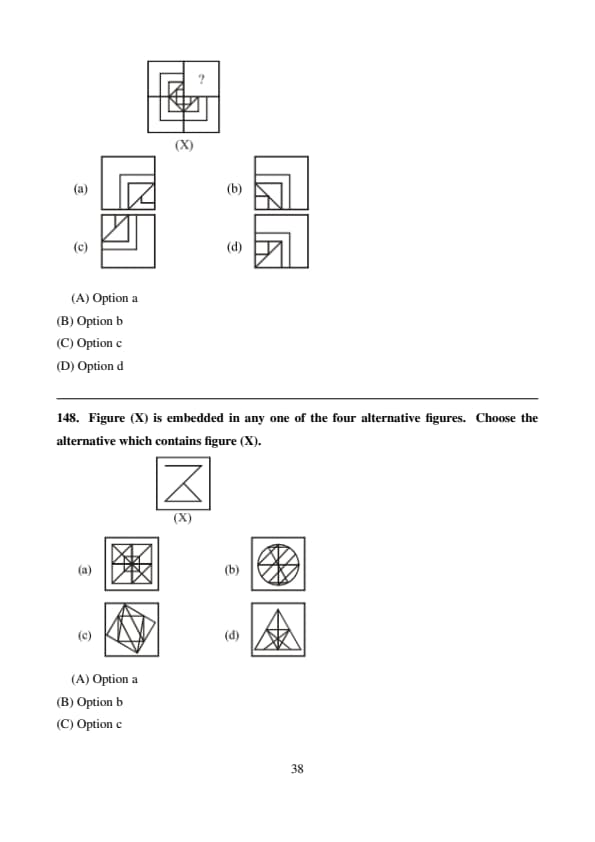
Figure (X) is embedded in any one of the four alternative figures. Choose the alternative which contains figure (X).

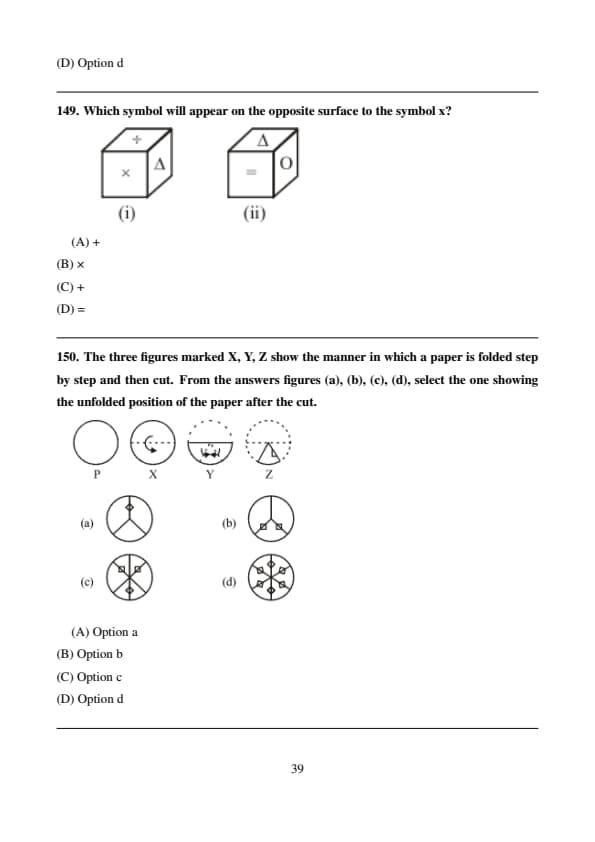
Which symbol will appear on the opposite surface to the symbol x?

The three figures marked X, Y, Z show the manner in which a paper is folded step by step and then cut. From the answers figures (a), (b), (c), (d), select the one showing the unfolded position of the paper after the cut.












































Comments