The JNTU Hyderabad will conduct the TS EAMCET 2025 Engineering Exam on May 4th, 2025, between 3:00 P.M. and 6:00 P.M.
Candidates appearing in this Shift can expect the TS EAMCET Question Paper to follow the standard format of 160 questions, divided into Mathematics (80), Physics (40), and Chemistry (40).
The Exam will be held in online mode with all questions being multiple choice and of 1 mark each, without any negative marking.
The TS EAMCET 2025 May 4th Shift 2 Question Paper with solution PDF is available here.
TS EAMCET 2025 Engineering May 4 Shift 2 Question Paper with Solution PDF
| TS EAMCET 2025 Engineering May 4 Shift 2 Question Paper | Download PDF | Check Solution |

The domain and range of \(f(x) = \frac{1}{\sqrt{|x|-x^2}}\) are A and B respectively. Then \(A \cup B = \) ?
A function \(f: \mathbb{R} \to \mathbb{R}\) defined by \(f(x) = \begin{cases} 2x+3, & x \le 4/3
-3x^2+8x, & x > 4/3 \end{cases}\) is
If \(2^{4n+3} + 3^{3n+1}\) is divisible by P for all natural numbers \(n\), then P is
A is a \(3 \times 3\) matrix satisfying \(A^3 - 5A^2 + 7A + I = 0\). If \(A^5 - 6A^4 + 12A^3 - 6A^2 + 2A + 2I = lA + mI\), then \(l + m =\)
If \(A = \begin{pmatrix} 0 & 1 & 2 \\
1 & 2 & 3 \\
3 & x & 1 \end{pmatrix}\), \(A^{-1} = \frac{1}{2} \begin{pmatrix} 1 & -1 & 1 \\
-8 & 6 & 2y \\
5 & -3 & 1 \end{pmatrix}\) then the point \((x,y)\) lies on the curve
Consider a homogeneous system of three linear equations in three unknowns represented by \(AX=O\). If \(X = \begin{pmatrix} l \\
m
0 \end{pmatrix}, l \neq 0, m \neq 0, l, m \in \mathbb{R}\) represents an infinite number of solutions of this system, then rank of A is
The number of real values of 'a', for which the system of equations \(2x+3y+az = 0\), \(x+ay-2z=0\) and \(3x+y+3z = 0\) has nontrivial solutions is
If the eight vertices of a regular octagon are given by the complex numbers \(\frac{1}{x_j-2i}\) (\(j=1,2,3,4,5,6,7,8\)), then the radius of the circumcircle of the octagon is
If \(|Z_1 - 3 - 4i| = 5\) and \(|Z_2| = 15\) then the sum of the maximum and minimum values of \(|Z_1 - Z_2|\) is
If \(Z=r(\cos\theta+i\sin\theta)\), \((\theta \neq -\pi/2)\) is a solution of \(x^3 = i\), then \(r^9(\cos(9\theta)+i\sin(9\theta)) =\)
If \(\omega \neq 1\) is a cube root of unity, then one root among the \(7^{th}\) roots of \((1+\omega)\) is
If \(f(x) = x^2 - 2(4K-1)x + g(K) > 0\) \(\forall x \in \mathbb{R}\) and for \(K \in (a,b)\), and if \(g(K) = 15K^2 - 2K - 7\), then
If local maximum of \(f(x) = \frac{ax+b}{(x-1)(x-4)}\) exists at \((2,-1)\), then \(a+b=\)
If \(1+2i\) is a root of the equation \(x^4 - 3x^3 + 8x^2 - 7x + 5 = 0\), then sum of the squares of the other roots is
If \(\alpha, \beta, \gamma\) are the roots of the equation \(x^3 + \frac{a}{2}x + b = 0\) and \((\alpha-\beta)(\alpha-\gamma)\), \((\beta-\alpha)(\beta-\gamma)\), \((\gamma-\alpha)(\gamma-\beta)\) are the roots of the equation \((y+a)^3 + K(y+a)^2 + L = 0\), then \(\frac{L}{K}= \)
All the letters of the word MOTHER are arranged in all possible ways and the resulting words (may or may not have meaning) are arranged as in the dictionary. The number of words that appear after the word MOTHER is
The number of positive integral solutions of \(\frac{1}{x} + \frac{1}{y} = \frac{1}{2025}\) is
The number of positive integral solutions of \(xyz = 60\) is
Numerically greatest term in the expansion of \((3x-4y)^{23}\) when \(x=\frac{1}{6}\) and \(y=\frac{1}{8}\) is
Let K be the number of rational terms in the expansion of \((\sqrt{2}+\sqrt[6]{3})^{6144}\). If the coefficient of \(x^P (P \in N)\) in the expansion of \(\frac{1}{(1+x)(1+x^2)(1+x^4)(1+x^8)(1+x^{16})}\) is \(a_P\), then \(a_K - a_{K+1} - a_{K-1} =\)
If \(\frac{3x+1}{(x-1)^2(x^2+1)} = \frac{A}{x-1} + \frac{B}{(x-1)^2} + \frac{Cx+D}{x^2+1}\), then \(2(A-C+B+D)=\)
If \(\tan(\frac{\pi}{4}+\frac{\alpha}{2}) = \tan^3(\frac{\pi}{4}+\frac{\beta}{2})\), then \(\frac{3+\sin^2\beta}{1+3\sin^2\beta}=\)
If \(P = \sin\frac{2\pi}{7} + \sin\frac{4\pi}{7} + \sin\frac{8\pi}{7}\) and \(Q = \cos\frac{2\pi}{7} + \cos\frac{4\pi}{7} + \cos\frac{8\pi}{7}\), then the point (P,Q) lies on the circle of radius
If \(\cos\alpha = \frac{l\cos\beta+m}{l+m\cos\beta}\), then \(\frac{\tan^2(\alpha/2)}{\tan^2(\beta/2)} =\)
If a, b are real numbers and \(\alpha\) is a real root of \(x^2+12+3\sin(a+bx)+6x=0\) then the value of \(\cos(a+b\alpha)\) for the least positive value of \(a+b\alpha\) is
The number of real solutions of \(\tan^{-1}x + \tan^{-1}(2x) = \frac{\pi}{4}\) is
Consider the following statements
Statement-I: \(\cosh^{-1}x = \tanh^{-1}x\) has no solution
Statement-II: \(\cosh^{-1}x = \coth^{-1}x\) has only one solution
The correct answer is
If the angular bisector of the angle A of the triangle ABC meets its circumcircle at E and the opposite side BC at D, then \(DE\cos\frac{A}{2} =\)
In a triangle ABC, \(a=5, b=4\) and \(\tan\frac{C}{2} = \sqrt{\frac{7}{9}}\), then its inradius r =
Two adjacent sides of a triangle are represented by the vectors \(2\vec{i}+\vec{j}-2\vec{k}\) and \(2\sqrt{3}\vec{i}-2\sqrt{3}\vec{j}+\sqrt{3}\vec{k}\). Then the least angle of the triangle and perimeter of the triangle are respectively
A plane \(\pi_1\) contains the vectors \(\vec{i}+\vec{j}\) and \(\vec{i}+2\vec{j}\). Another plane \(\pi_2\) contains the vectors \(2\vec{i}-\vec{j}\) and \(3\vec{i}+2\vec{k}\). \(\vec{a}\) is a vector parallel to the line of intersection of \(\pi_1\) and \(\pi_2\). If the angle \(\theta\) between \(\vec{a}\) and \(\vec{i}-2\vec{j}+2\vec{k}\) is acute, then \(\theta=\)
In a quadrilateral ABCD, \(\angle A = \frac{2\pi}{3}\) and AC is the bisector of angle A. If \(15|AC| = 5|AD| = 3|AB|\), then the angle between \(\vec{AB}\) and \(\vec{BC}\) is
\(\vec{a}, \vec{b}, \vec{c}\) are three non-coplanar and mutually perpendicular vectors of same magnitude K. \(\vec{r}\) is any vector satisfying \(\vec{a}\times((\vec{r}-\vec{b})\times\vec{a}) + \vec{b}\times((\vec{r}-\vec{c})\times\vec{b}) + \vec{c}\times((\vec{r}-\vec{a})\times\vec{c}) = \vec{0}\), then \(\vec{r} =\)
Consider the following
Assertion (A): The two lines \(\vec{r} = \vec{a}+t(\vec{b})\) and \(\vec{r}=\vec{b}+s(\vec{a})\) intersect each other.
Reason (R): The shortest distance between the lines \(\vec{r}=\vec{p}+t(\vec{q})\) and \(\vec{r}=\vec{c}+s(\vec{d})\) is equal to the length of projection of the vector \((\vec{p}-\vec{c})\) on \((\vec{q}\times\vec{d})\).
The correct answer is
The mean deviation about median of the numbers \(3x, 6x, 9x, ..., 81x\) is 91, then \(|x|=\)
Functions are formed from the set \(A = \{a_1, a_2, a_3\}\) to another set \(B = \{b_1, b_2, b_3, b_4, b_5\}\). If a function is selected at random, the probability that it is a one-one function is
A and B are two events of a random experiment such that \(P(B)=0.4\), \(P(A \cap \bar{B}) = 0.5\), \(P(A \cup B) + P(A|B) = 1.15\), then \(P(A)=\)
There are two boxes each containing 10 balls. In each box, few of them are black balls and rest are white. A ball is drawn at random from one of the boxes and found that it is black. If the probability that the black ball drawn is from the second box is \(\frac{1}{5}\), then number of black balls in the first box is
In a shelf there are three mathematics and two physics books. A student takes a book randomly. If he randomly takes, successively for three times by replacing the book already taken every time, then the mean of the number of mathematics books which is treated as random variable is
In a Poisson distribution, if \(\frac{P(X=5)}{P(X=2)} = \frac{1}{7500}\) and \(\frac{P(X=5)}{P(X=3)} = \frac{1}{500}\), then the mean of the distribution is
A(2,0), B(0,2), C(-2,0) are three points. Let a, b, c be the perpendicular distances from a variable point P on to the lines AB, BC and CA respectively. If a, b, c are in arithmetic progression, then the locus of P is
When the coordinate axes are rotated about the origin through an angle \(\frac{\pi}{4}\) in the positive direction, the equation \(ax^2+2hxy+by^2=c\) is transformed to \(25x'^2+9y'^2=225\), then \((a+2h+b-\sqrt{c})^2=\)
\(y-x=0\) is the equation of a side of a triangle ABC. The orthocentre and circumcentre of the triangle ABC are respectively (5,8) and (2,3). The reflection of orthocentre with respect to any side of the triangle lies on its circumcircle. Then the radius of the circumcircle of the triangle is
Two families of lines are given by \(ax+by+c=0\) and \(4a^2+9b^2-c^2-12ab=0\). Then the line common to both the families is
Two non parallel sides of a rhombus are parallel to the lines \(x+y-1=0\) and \(7x-y-5=0\). If (1,3) is the centre of the rhombus and one of its vertices \(A(\alpha, \beta)\) lies on \(15x-5y=6\), then one of the possible values of \((\alpha+\beta)\) is
If the equations \(3x^2+2hxy-3y^2=0\) and \(3x^2+2hxy-3y^2+2x-4y+c=0\) represent the four sides of a square, then \(\frac{h}{c}= \)
The radius of the circle having three chords along y-axis, the line \(y=x\) and the line \(2x+3y=10\) is
Among the chords of the circle \(x^2+y^2=75\), the number of chords having their midpoints on the line \(x=8\) and having their slopes as integers is
The equation of the circle which touches the circle \(S \equiv x^2+y^2-10x-4y+19=0\) at the point (2,3) internally and having radius equal to half of the radius of the circle S=0 is
If \(P(\frac{7}{5}, \frac{6}{5})\) is the inverse point of \(A(1,2)\) with respect to a circle with centre \(C(2,0)\), then the radius of that circle is
If the circle \(S=0\) intersect the three circles \(S_1 = x^2+y^2+4x-7=0\), \(S_2 = x^2+y^2+y=0\) and \(S_3 = x^2+y^2+\frac{3}{2}x+\frac{5}{2}y-\frac{9}{2}=0\) orthogonally, then the radical axis of \(S=0\) and \(S_1=0\) is
If a tangent to the circle \(x^2+y^2+2x+2y+1=0\) is radical axis of the circles \(x^2+y^2+2gx+2fy+c=0\) and \(2x^2+2y^2+3x+8y+2c=0\), then
If the angle between the tangents drawn to the parabola \(y^2=4x\) from the points on the line \(4x-y=0\) is \(\frac{\pi}{3}\), then the sum of the abscissae of all such points is
The normal at a point on the parabola \(y^2=4x\) passes through a point P. Two more normals to this parabola also pass through P. If the centroid of the triangle formed by the feet of these three normals is G(2,0), then the abscissa of P is
The circumcenter of the equilateral triangle having the three points \(\theta_1, \theta_2, \theta_3\) lying on the ellipse \(\frac{x^2}{a^2}+\frac{y^2}{b^2}=1\) as its vertices is \((r,s)\). Then the average of \(\cos(\theta_1-\theta_2), \cos(\theta_2-\theta_3)\) and \(\cos(\theta_3-\theta_1)\) is
The ellipse \(\frac{x^2}{a^2}+\frac{y^2}{b^2}=1\) (\(b>a\)) is an ellipse with eccentricity \(\frac{1}{\sqrt{2}}\). If the angle of intersection between the ellipse and parabola \(y^2=4ax\) is \(\theta\), then the coordinates of the point \(\frac{20}{3}\) on the ellipse is
The number of common tangents that can be drawn to the curves \(\frac{x^2}{16}-\frac{y^2}{9}=1\) and \(x^2+y^2=16\) is
Let A(\(\alpha\),4,7) and B(3,\(\beta\),8) be two points in space. If YZ plane and ZX plane respectively divide the line segment joining the points A and B in the ratio 2:3 and 4:5, then the point C which divides AB in the ratio \(\alpha:\beta\) externally is
The direction ratios of the line bisecting the angle between the x-axis and the line having direction ratios (3, -1, 5) are
If the plane \(-4x-2y+2z+\alpha=0\) is at a distance of two units from the plane \(2x+y-z+1=0\), then the product of all the possible values of \(\alpha\) is
\(\lim_{x \to 0} \frac{\sqrt[3]{\cos x} - \sqrt{\cos x}}{\sin^2 x} =\)
Let \(f:[-1,2] \to \mathbb{R}\) be defined by \(f(x) = [x^2-3]\) where \([.]\) denotes greatest integer function, then the number of points of discontinuity for the function \(f\) in \((-1,2)\) is
If \(f(x) = \begin{cases} x^2 \cos(\frac{\pi}{x}), & x \neq 0
0, & x=0 \end{cases}\), then at \(x=0\), \(f(x)\) is
(Note: The question image says 'at \(x=2\)', but this is a standard problem about the point \(x=0\). At \(x=2\), the function is clearly differentiable by standard rules. We assume the question meant \(x=0\).)
The set of all values of x for which \(f(x) = ||x|-1|\) is differentiable is
If \(y=f(x)^{g(x)}\) and \(\frac{dy}{dx} = y[H(x)f'(x)+G(x)g'(x)]\), then \(\int \frac{G(x)H(x)f'(x)}{g(x)}dx =\)
If \(x=t-\sin t, y=1-\cos t\) and \(\frac{d^2y}{dx^2}=-1\) at \(t=K, K>0\), then \(\lim_{t \to K} \frac{y}{x} =\)
For the curve \((\frac{x}{a})^n + (\frac{y}{b})^n = 2\), (\(n \in N\) \& \(n>1\)) the line \(\frac{x}{a}+\frac{y}{b}=2\) is
The height of a cone with semi vertical angle \(\pi/3\) is increasing at the rate of 2 units/min. The rate at which the radius of the cone is to be decreased so as to have a fixed volume always is
The function \(f(x)=2x^3-9ax^2+12a^2x+1\) where \(a>0\) attains its local maximum and local minimum at p and q respectively. If \(p^2=q\) then a =
Consider all functions given in List-I in the interval [1,3]. The List-2 has the values of 'c' obtained by applying Lagrange's mean value theorem on the functions of List-1. Match the functions and values of 'c'.

If the percentage error in the radius of a circle is 3, then the percentage error in its area is
If \(I_1 = \int \frac{e^x}{e^{4x}+e^{2x}+1}dx\), \(I_2 = \int \frac{e^{-x}}{e^{-4x}+e^{-2x}+1}dx\), then \(I_2-I_1=\)
If \(\int \frac{\sqrt{1-\sqrt{x}}}{\sqrt{x(1+\sqrt{x})}}dx = 2f(x)-2\sin^{-1}\sqrt{x}+c\), then \(f(x)=\)
If \(\int \frac{3x+2}{4x^2+4x+5}dx = A\log(4x^2+4x+5)+B\tan^{-1}(\frac{2x+1}{2})+c\), then \(A+B=\)
Consider the following
Assertion (A): \(\int \sqrt{x-3}(\sin^{-1}(\log x) + \cos^{-1}(\log x))dx = \frac{\pi}{3}(x-3)^{3/2}+c\)
Reason (R): \(\sin^{-1}(f(x))+\cos^{-1}(f(x))=\frac{\pi}{2}\), \(|f(x)|\le 1\)
The correct answer is
\(\lim_{n \to \infty} \frac{(2n(2n-1)...(n+2)(n+1))^{1/n}}{n} =\)
The area of the region bounded by \(y=x^3\), x-axis, \(x=-2\) and \(x=4\) is
If \(\int_0^{\pi/2} \tan^{14}(x/2)dx = 2\left[\sum_{n=1}^7 f(n) - \frac{\pi}{4}\right]\), then \(f(n)=\)
The differential equation of the family of all circles of radius 'a' is
If the general solution of \((1+y^2)dx = (\tan^{-1}y - x)dy\) is \(x = f(y)+ce^{-\tan^{-1}y}\), then \(f(y)=\)
The force of mutual attraction between any two objects by virtue of their masses is
The error in the measurement of force acting normally on a square plate is 3%. If the error in the measurement of the side of the plate is 1%, then the error in the determination of the pressure acting on the plate is
For a particle moving along a straight line path, the displacements in third and fifth seconds of its motion are 10 m and 18 m respectively. The speed of the particle at time t=4s is
The vertical displacement (y in metre) of a projectile in terms of its horizontal displacement (x in metre) is given by \(y=(\sqrt{3}x - 0.2x^2)\). The time of flight of the projectile is (Acceleration due to gravity = 10 ms\(^{-2}\))
A block of mass \(\sqrt{2}\) kg is placed on a rough horizontal surface. A force 'F' acting upwards at an angle of 45\(^\circ\) with the horizontal causes the block to start motion. If the coefficient of static friction between the surface and the block is 0.25, the magnitude of the force 'F' is (Acceleration due to gravity = 10 ms\(^{-2}\))
If the kinetic energy of a body moving with a velocity of \((2\vec{i}+3\vec{j}-4\vec{k})\) ms\(^{-1}\) is 87 J, then the mass of the body is
A body of mass 0.5 kg is supplied with a power 'P' (in watt) which varies with time 't' (in second) as \(P=3t^2+3\). If the velocity of the body at time t=0 is zero, then the velocity of the body at time t=3s is
A solid sphere of mass 2 kg and radius 0.5 m is rolling without slipping on a horizontal surface. The ratio of the rotational and translational kinetic energies of the sphere is
If the length of a thin uniform rod is 'L' and the radius of gyration of the rod about an axis perpendicular to its length and passing through one end is K, then K:L=
The force (F in newton) acting on a particle of mass 90 g executing simple harmonic motion is given by \(F+0.04\pi^2y=0\), where y is displacement of the particle in meter. If the amplitude of the particle is \(\frac{6}{\pi}\) m, then the maximum velocity of the particle is
Which of the following is incorrect about the gravitational force between two bodies?
A steel rod with a circular cross-section of diameter 1cm and another steel rod with a square cross-section of side 1cm have equal mass. If the two rods are subjected to same tension, the ratio of the elongations of the two rods is
A cube of side 40 cm is floating with \(\frac{1}{4}\)th of its volume immersed in water. When a circular disc is placed on the cube, it floats with \(\frac{2}{5}\)th of its volume immersed in water. The mass of the disc is
The maximum length of water column that can stay without falling in a vertically held capillary tube of diameter 1mm and open at both the ends is (Acceleration due to gravity = 10 ms\(^{-2}\) and surface tension of water = 0.07 Nm\(^{-1}\))
A steel pendulum clock manufactured at 32\(^\circ\)C and working at 47\(^\circ\)C is nearly (Coefficient of linear expansion of steel = \(12 \times 10^{-6}\) /\(^\circ\)C)
A metal metre scale that is accurate up to 0.5 mm is made at a temperature of 25\(^\circ\)C. The range of temperatures within which it can be used is (Coefficient of linear expansion of the metal = \(10^{-5}\) /\(^\circ\)C)
A Carnot engine uses diatomic gas as a working substance. During the adiabatic expansion part of the cycle, if the volume of the gas becomes 32 times its initial volume, then the efficiency of the engine is
The ratio of the average translational kinetic energies of hydrogen and oxygen at the same temperature is
The air columns in two tubes closed at one end vibrating in their fundamental modes produce 2 beats per second. The number of beats produced per second when the same tubes are vibrated in their fundamental mode with their both ends open are
A car moving towards a cliff emits sound of frequency 'n'. If the difference in frequencies of the horn and its echo heard by the driver of the car is 10% of 'n', then the speed of the car is nearly (Speed of sound in air is 336ms\(^{-1}\))
A straight metal rod of length 6 cm is placed along the principal axis of a concave mirror of focal length 9 cm such that the end of the rod closer to the mirror is at a distance of 15 cm from the pole of the mirror. The length of the image of the rod is
A ray of light incidents at an angle of \(9.3^\circ\) on one face of a small angle prism of refracting angle \(6^\circ\). If the ray of light emerges normally from the second face, the refractive index of the material of the prism is
The distance for which ray optics becomes a good approximation for an aperture of 0.3 cm and a light of wavelength 6000 \(\AA\) is
The electrostatic force between two charges kept in air is F. If 30% of the space between the charges is filled with a medium, then the electrostatic force between the charges becomes \(\frac{F}{2.56}\). The dielectric constant of the medium is
729 small identical spheres each charged to an electric potential 3V combine to form a bigger sphere. The electric potential of the bigger sphere is
The current amplification factor of a transistor in common emitter configuration is 80. If the emitter current is 2.43 mA, then the base current is
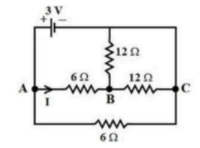
The area of cross-section of a potentiometer wire is \(6 \times 10^{-7}\) m\(^2\). The potential difference per unit length of the potentiometer wire when it is connected to a cell of negligible internal resistance and a resistor in series is \(0.15\) Vm\(^{-1}\). If the current through potentiometer wire is \(0.3\)A, then the resistivity of the material of the potentiometer wire is
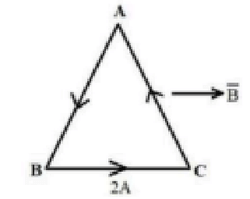
As shown in the figure, a uniform straight wire of length \(30\sqrt{3}\) cm is bent in the form of an equilateral triangle ABC. A uniform magnetic field 2T is applied parallel to the side BC. If the current through the wire is 2A, the magnitude of the force on the side AC is
A proton moving with a velocity of \(8\times 10^5\) ms\(^{-1}\) enters a uniform magnetic field normal to the direction of the magnetic field. If the radius of the circular path of the proton in the magnetic field is \(8.3\) cm, then the magnitude of the magnetic field is (Charge of proton = \(1.6\times 10^{-19}\) C and mass of the proton = \(1.66\times 10^{-27}\) kg)
At a certain place in the magnetic meridian, the earth's magnetic field is twice its vertical component. The ratio of horizontal component of earth's magnetic field and the total magnetic field of the earth at that place is
A coil of resistance \(16 \Omega\) is placed with its plane perpendicular to a uniform magnetic field whose flux (\(\phi\) in \(10^{-3}\) weber) changes with time (t in second) as \(\phi = 5t^2+4t+2\). The induced current at time \(t=6\) seconds is
The small energy losses in transformers due to eddy currents can be reduced by
If the electric field of a plane electromagnetic wave is \(E_z = 60\sin(0.5\times 10^3x + 1.5\times 10^{11}t)\) Vm\(^{-1}\), then the magnetic field of the wave is
In a photoelectric experiment, the slope of the graph drawn between stopping potential along y-axis and frequency of incident radiation along x-axis is (Planck's constant = \(6.6\times 10^{-34}\) Js)
The maximum wavelength of incident radiation required to ionize a hydrogen atom in its ground state is nearly
When an element \(_{90}^{232}Th\) decays into \(_{82}^{208}Pb\), the number of \(\alpha\) and \(\beta^{-}\) particles emitted respectively are
During the disintegration of a radioactive nucleus of mass number 208 at rest, two alpha particles each with kinetic energy E are emitted. The total kinetic energy of the emitted alpha particles and the daughter nucleus after the disintegration is
The current amplification factor of a transistor in common emitter configuration is 80. If the emitter current is 2.43 mA, then the base current is
The negative feedback in an amplifier
If the frequencies of the carrier wave and message signal are \(1\) MHz and \(28\) kHz respectively, then the frequencies of the side bands are
Observe the following statements
Statement-I: Rutherford model of an atom cannot explain the stability of an atom
Statement-II: The wavelength of X-rays is higher than the wavelength of microwaves
The correct answer is
In hydrogen atom, an electron is transferred from an orbit of radius \(1.3225\) nm to another orbit of radius \(0.2116\) nm. What is the energy (in J) of emitted radiation? (Rydberg constant \(R_H \approx 1.097 \times 10^7 m^{-1}\))
Identify the correct orders regarding atomic radii
i. \(Cl > F > Li\)
ii. \(P > C > N\)
iii. \(Tm > Sm > Eu\)
iv. \(Sr > Ca > Mg\)
Match the following
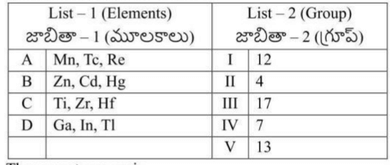
The atomic numbers of the elements X, Y, Z are \(a, a+1, a+2\) respectively. Z is an alkali metal. The nature of bonding in the compound formed by X and Z is
The sets of molecules in which central atom has no lone pair of electrons are:
i. \(SnCl_2, NH_3, SF_4\)
ii. \(HgCl_2, SO_3, SF_6\)
iii. \(BeCl_2, BF_3, PCl_5\)
iv. \(ClF_3, BrF_5, XeF_6\)
The isobars of one mole of an ideal gas were obtained at three different pressures \((p_1, p_2\) and \(p_3)\). The slopes of these isobars are \(m_1, m_2\) and \(m_3\) respectively. If \(p_1 < p_2 < p_3\), then the correct relation of the slopes is
\(100 mL\) of \(0.05 M Cu^{2+}\) aqueous solution is added to \(1 L\) of \(0.1 M KI\) solution. The number of moles of \(I_2\) and \(Cu_2I_2\) formed are respectively
The \(C_p\) of an ideal gas is \(10.314 J mol^{-1} K^{-1}\). One mole of this gas is expanded against a constant pressure of \(p atm\). The change in temperature during expansion is \(1.0 K\). The values of \(q\) (in J) and \(\Delta H\) (in \(J mol^{-1}\)) are respectively
At T(K), \(K_p\) value for the reaction, \(2AO_2(g)+O_2(g) \rightleftharpoons 2AO_3(g)\) is \(4\times 10^{10}\). What is the \(K'_p\) value for \(3AO_2(g)+\frac{3}{2}O_2(g) \rightleftharpoons 3AO_3(g)\) at T(K)?
A sample of water contains \(Mg(HCO_3)_2\) and \(Ca(HCO_3)_2\). On boiling this water, these hydrogen carbonates are removed as precipitates. The precipitates are
Which of the following statements is not correct?
The order of negative standard potential values of \(Li, Na, K\) is
In which of the following reactions, hydrogen is evolved?
I. \(Reaction of sodium borohydride with iodine\)
II. \(Oxidation of diborane\)
III. \(Reaction of boron trifluoride with sodium hydride\)
IV. \(Hydrolysis of diborane\)
Which of the following statements is not correct regarding the gas evolved by the reaction of dilute \(HCl\) on \(CaCO_3\)?
Observe the following statements
Statement-I: The carbon containing components of photochemical smog are acrolein, methanal and PAN
Statement-II: The number of greenhouse gases in the list given below is 5
\(CH_4, CO_2, NO, H_2O(l), H_2O(g), O_2, O_3\)
The correct answer is
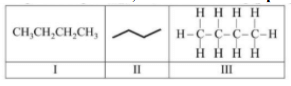
The condensed, bond line and complete formulae of n-butane are respectively
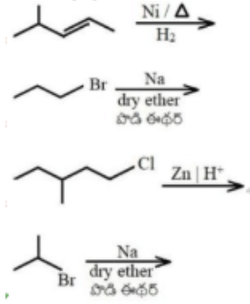
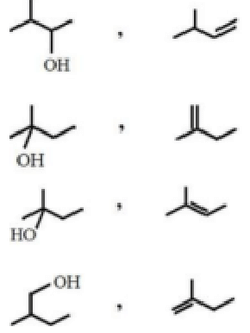
'X' is the isomer of \(C_6H_{14}\). It has four primary carbons and two tertiary carbons. 'X' can be prepared from which of the following reaction?
What are X and Y respectively in the following reaction sequence?
\[ Iso-pentane \xrightarrow{KMnO_4} X \xrightarrow{\substack{20%\,H_3PO_4
358\,K}} Y \]
What are X and Y respectively in the following reaction sequence?
A metal (M), crystallizes in \(fcc\) lattice with edge length of \(4.242 A\). What is the radius of \(M\) atom (in \(A\))?
A solid mixture weighing \(5 g\) contains equal number of moles of \(Na_2CO_3\) and \(NaHCO_3\). This solid mixture was dissolved in \(1 L\) of water. What is the volume (in \(mL\)) of \(0.1 M HCl\) required to completely react with this \(1 L\) mixture solution?
At \(298 K\) the equilibrium constant for the reaction, \(M(s)+2Ag^+(aq) \rightleftharpoons M^{2+}(aq)+2Ag(s)\) is \(10^{15}\). What is the \(E_{cell}^{\circ}\) (in \(V\)) for this reaction? (\(\frac{2.303 RT}{F} = 0.06 V\))
\(A\to P\) is a first order reaction. At \(300 K\) this reaction was started with \([A]=0.5 molL^{-1}\). The rate constant of reaction was \(0.125 min^{-1}\). The same reaction was started separately with \([A]=1 molL^{-1}\) at \(300 K\). The rate constant (in \(min^{-1}\)) now is
Observe the following reactions
I. \(CO(g)+H_2(g) \xrightarrow{X} HCHO(g)\)
II. \(CO(g)+3H_2(g) \xrightarrow{Y} CH_4(g)+H_2O(g)\)
The catalysts \(X\) and \(Y\) in the above reactions are respectively
Composition of siderite ore is
Which of the following gives more number of oxides on reacting with \(HCl\)?
The number of lone pairs of electrons on the central atom of \(XeO_3, XeOF_4\) and \(XeF_6\) respectively is
Which of the following statements is not correct?
The pair of ions with paramagnetic nature and same number of electrons is
Observe the following complex ions Identify the option in which the unpaired electrons in the complex ions are in correct increasing order

The polymer chains are held together by hydrogen bonding in a polymer X. Polymer X is formed from monomers Y and Z. What are Y and Z?
![]()
Amino acid 'X' contains phenolic hydroxy group and amino acid 'Y' contains amide group. 'X' and 'Y' respectively are
The chemical X is used in the prevention of heart attack. The structure of X is
Observe the following reactions reaction is
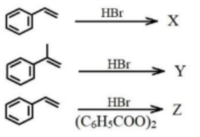
Consider the following set of reactions.
\(C_6H_5CN \xrightarrow{B} Y \xrightarrow{A} X (reacts with 2,4-DNP)\)
\(C_6H_5CN \xrightarrow{A} X (dissolves in dil. HCl)\)
What are A and B respectively?
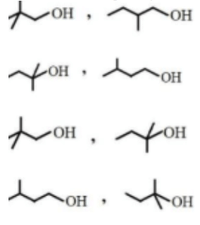
An alcohol X (\(C_5H_{12}O\)) produces turbidity instantly with conc. \(HCl/ZnCl_2\). Isomer (Y) of \(X\) undergoes dehydration with conc. \(H_2SO_4\) at \(443 K\). \(X\) and \(Y\) respectively are
What are \(X\) and \(Y\) in the following reaction sequence?
\(C_5H_{12}O \xrightarrow{Cu/573 K} C_5H_{10} \xrightarrow{(i) O_3, (ii) Zn+H_2O} X + Y\)
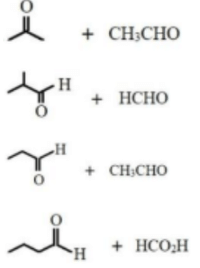
Observe the following set of reactions
{l} \(C_6H_5COCl \xrightarrow{X} C_6H_5CHO\)
\(C_6H_5COCl \xrightarrow{Y} C_6H_5COCH_3\)
\(C_6H_5CHO \xrightarrow{OH^-/293 K} Z (Major product)\)
What are \(X, Y\) and \(Z\) respectively?
View Solution
Step 1: Identify reagent X (Reduction of acyl chloride to aldehyde).
The reaction is: \[ C_6H_5COCl \xrightarrow{X} C_6H_5CHO \]
This is the partial reduction of an acyl chloride to an aldehyde.
Reagent \(X\) must be \(H_2/Pd-BaSO_4\) (Rosenmund reduction).
Hence, \(\boxed{X = H_2/Pd-BaSO_4}\)
Step 2: Identify reagent Y (Formation of ketone).
The reaction: \[ C_6H_5COCl \xrightarrow{Y} C_6H_5COCH_3 \]
This can be done using a dialkyl cadmium reagent \((CH_3)_2Cd\) or a Grignard reagent \((CH_3MgBr)\).
Though organocadmium is more specific, the question provides \(CH_3MgBr\) as the option, so we take: \[ \boxed{Y = CH_3MgBr} \]
Step 3: Identify product Z.
The reaction is: \[ C_6H_5CHO \xrightarrow{OH^-/293\,K} Z \]
Under basic conditions, Benzaldehyde can undergo condensation reactions. If the product is given as \(C_6H_5CH=CH-C_6H_5\) (Stilbene), it indicates an aldol-type or Wittig-type condensation product.
Hence, \(\boxed{Z = C_6H_5CH=CH-C_6H_5}\) \; (Stilbene).
Step 4: Final Answer Summary.
\[ \text{X = H_2/Pd-BaSO_4, \quad Y = CH_3MgBr, \quad Z = C_6H_5CH=CH-C_6H_5 \] Quick Tip: The Rosenmund reduction (\(H_2/Pd-BaSO_4\)) is a name reaction for the partial reduction of acyl chlorides to aldehydes. Organometallic reagents like \(R_2Cd\) are best for ketone synthesis. Aldol condensation of aromatic aldehydes with base (like \(C_6H_5CHO\)) is typically a Cannizzaro reaction, unless a strong nucleophile is added.
Consider the following set of reactions.
\(C_6H_5CN \xrightarrow{B} Y (reacts with 2,4-DNP) \xrightarrow{A} X (dissolves in dil. HCl)\)
What are A and B respectively?













































TS EAMCET 2025 Expected Difficulty Level
Based on the analysis of past shifts and expert opinions, the TS EAMCET 2025 Question Paper for the Engineering exam, May 4th 2025 Shift 2, is expected to be of a moderate difficulty level.
| Subject | Expected Difficulty Level | Key Observations |
| Mathematics | Moderate to Tough | It is expected to have Lengthy calculation-based questions with emphasis on vectors, calculus, and coordinate geometry |
| Physics | Moderate | It is expected to have Conceptually based numericals with a focus on thermodynamics, optics, and electrodynamics |
| Chemistry | Easy to Moderate | It will be mostly NCERT-based, with questions from chemical bonding and organic chemistry |
| Overall | Moderate | Balanced paper with a mix of direct and application-based questions |
TS EAMCET 2025 Topic-Wise Weightage
The TS EAMCET 2025 Engineering Exam has a total of 160 multiple-choice questions that are allocated in the following pattern: Mathematics (80), Physics (40), and Chemistry (40).
Understanding of topic-wise weightage helps candidates to estimate the topics that can be asked in the TS EAMCET Question Paper.
Mathematics (80 Questions)
| Topic | Expected Questions | Approximate Weightage (%) |
|---|---|---|
| Algebra | 10–12 | 12% |
| Calculus | 12–14 | 15% |
| Coordinate Geometry | 10–12 | 12% |
| Trigonometry | 8–10 | 10% |
| Probability & Statistics | 8–10 | 10% |
| Vector Algebra | 6–8 | 8% |
Physics (40 Questions)
| Topic | Expected Questions | Approximate Weightage (%) |
|---|---|---|
| Mechanics | 10–12 | 25% |
| Thermodynamics | 6–8 | 15% |
| Electromagnetism | 6–8 | 15% |
| Optics | 4–6 | 10% |
| Modern Physics | 4–6 | 10% |
Chemistry (40 Questions)
| Topic | Expected Questions | Approximate Weightage (%) |
| Organic Chemistry | 12–14 | 30% |
| Inorganic Chemistry | 10–12 | 25% |
| Physical Chemistry | 12–14 | 30% |
The JNTU Hyderabad will conduct the TS EAMCET 2025 Engineering Exam on May 4th, 2025, between 3:00 P.M. and 6:00 P.M.
Candidates appearing in this Shift can expect the TS EAMCET Question Paper to follow the standard format of 160 questions, divided into Mathematics (80), Physics (40), and Chemistry (40).
The Exam will be held in online mode with all questions being multiple choice and of 1 mark each, without any negative marking.
The TS EAMCET 2025 May 4th Shift 2 Question Paper with solution PDF is available here.
TS EAMCET 2025 Engineering May 4 Shift 2 Question Paper with Solution PDF
| TS EAMCET 2025 Engineering May 4 Shift 2 Question Paper | Download PDF | Check Solution |

The domain and range of \(f(x) = \frac{1}{\sqrt{|x|-x^2}}\) are A and B respectively. Then \(A \cup B = \) ?
A function \(f: \mathbb{R} \to \mathbb{R}\) defined by \(f(x) = \begin{cases} 2x+3, & x \le 4/3
-3x^2+8x, & x > 4/3 \end{cases}\) is
If \(2^{4n+3} + 3^{3n+1}\) is divisible by P for all natural numbers \(n\), then P is
A is a \(3 \times 3\) matrix satisfying \(A^3 - 5A^2 + 7A + I = 0\). If \(A^5 - 6A^4 + 12A^3 - 6A^2 + 2A + 2I = lA + mI\), then \(l + m =\)
If \(A = \begin{pmatrix} 0 & 1 & 2 \\
1 & 2 & 3 \\
3 & x & 1 \end{pmatrix}\), \(A^{-1} = \frac{1}{2} \begin{pmatrix} 1 & -1 & 1 \\
-8 & 6 & 2y \\
5 & -3 & 1 \end{pmatrix}\) then the point \((x,y)\) lies on the curve
Consider a homogeneous system of three linear equations in three unknowns represented by \(AX=O\). If \(X = \begin{pmatrix} l \\
m
0 \end{pmatrix}, l \neq 0, m \neq 0, l, m \in \mathbb{R}\) represents an infinite number of solutions of this system, then rank of A is
The number of real values of 'a', for which the system of equations \(2x+3y+az = 0\), \(x+ay-2z=0\) and \(3x+y+3z = 0\) has nontrivial solutions is
If the eight vertices of a regular octagon are given by the complex numbers \(\frac{1}{x_j-2i}\) (\(j=1,2,3,4,5,6,7,8\)), then the radius of the circumcircle of the octagon is
If \(|Z_1 - 3 - 4i| = 5\) and \(|Z_2| = 15\) then the sum of the maximum and minimum values of \(|Z_1 - Z_2|\) is
If \(Z=r(\cos\theta+i\sin\theta)\), \((\theta \neq -\pi/2)\) is a solution of \(x^3 = i\), then \(r^9(\cos(9\theta)+i\sin(9\theta)) =\)
If \(\omega \neq 1\) is a cube root of unity, then one root among the \(7^{th}\) roots of \((1+\omega)\) is
If \(f(x) = x^2 - 2(4K-1)x + g(K) > 0\) \(\forall x \in \mathbb{R}\) and for \(K \in (a,b)\), and if \(g(K) = 15K^2 - 2K - 7\), then
If local maximum of \(f(x) = \frac{ax+b}{(x-1)(x-4)}\) exists at \((2,-1)\), then \(a+b=\)
If \(1+2i\) is a root of the equation \(x^4 - 3x^3 + 8x^2 - 7x + 5 = 0\), then sum of the squares of the other roots is
If \(\alpha, \beta, \gamma\) are the roots of the equation \(x^3 + \frac{a}{2}x + b = 0\) and \((\alpha-\beta)(\alpha-\gamma)\), \((\beta-\alpha)(\beta-\gamma)\), \((\gamma-\alpha)(\gamma-\beta)\) are the roots of the equation \((y+a)^3 + K(y+a)^2 + L = 0\), then \(\frac{L}{K}= \)
All the letters of the word MOTHER are arranged in all possible ways and the resulting words (may or may not have meaning) are arranged as in the dictionary. The number of words that appear after the word MOTHER is
The number of positive integral solutions of \(\frac{1}{x} + \frac{1}{y} = \frac{1}{2025}\) is
The number of positive integral solutions of \(xyz = 60\) is
Numerically greatest term in the expansion of \((3x-4y)^{23}\) when \(x=\frac{1}{6}\) and \(y=\frac{1}{8}\) is
Let K be the number of rational terms in the expansion of \((\sqrt{2}+\sqrt[6]{3})^{6144}\). If the coefficient of \(x^P (P \in N)\) in the expansion of \(\frac{1}{(1+x)(1+x^2)(1+x^4)(1+x^8)(1+x^{16})}\) is \(a_P\), then \(a_K - a_{K+1} - a_{K-1} =\)
If \(\frac{3x+1}{(x-1)^2(x^2+1)} = \frac{A}{x-1} + \frac{B}{(x-1)^2} + \frac{Cx+D}{x^2+1}\), then \(2(A-C+B+D)=\)
If \(\tan(\frac{\pi}{4}+\frac{\alpha}{2}) = \tan^3(\frac{\pi}{4}+\frac{\beta}{2})\), then \(\frac{3+\sin^2\beta}{1+3\sin^2\beta}=\)
If \(P = \sin\frac{2\pi}{7} + \sin\frac{4\pi}{7} + \sin\frac{8\pi}{7}\) and \(Q = \cos\frac{2\pi}{7} + \cos\frac{4\pi}{7} + \cos\frac{8\pi}{7}\), then the point (P,Q) lies on the circle of radius
If \(\cos\alpha = \frac{l\cos\beta+m}{l+m\cos\beta}\), then \(\frac{\tan^2(\alpha/2)}{\tan^2(\beta/2)} =\)
If a, b are real numbers and \(\alpha\) is a real root of \(x^2+12+3\sin(a+bx)+6x=0\) then the value of \(\cos(a+b\alpha)\) for the least positive value of \(a+b\alpha\) is
The number of real solutions of \(\tan^{-1}x + \tan^{-1}(2x) = \frac{\pi}{4}\) is
Consider the following statements
Statement-I: \(\cosh^{-1}x = \tanh^{-1}x\) has no solution
Statement-II: \(\cosh^{-1}x = \coth^{-1}x\) has only one solution
The correct answer is
If the angular bisector of the angle A of the triangle ABC meets its circumcircle at E and the opposite side BC at D, then \(DE\cos\frac{A}{2} =\)
In a triangle ABC, \(a=5, b=4\) and \(\tan\frac{C}{2} = \sqrt{\frac{7}{9}}\), then its inradius r =
Two adjacent sides of a triangle are represented by the vectors \(2\vec{i}+\vec{j}-2\vec{k}\) and \(2\sqrt{3}\vec{i}-2\sqrt{3}\vec{j}+\sqrt{3}\vec{k}\). Then the least angle of the triangle and perimeter of the triangle are respectively
A plane \(\pi_1\) contains the vectors \(\vec{i}+\vec{j}\) and \(\vec{i}+2\vec{j}\). Another plane \(\pi_2\) contains the vectors \(2\vec{i}-\vec{j}\) and \(3\vec{i}+2\vec{k}\). \(\vec{a}\) is a vector parallel to the line of intersection of \(\pi_1\) and \(\pi_2\). If the angle \(\theta\) between \(\vec{a}\) and \(\vec{i}-2\vec{j}+2\vec{k}\) is acute, then \(\theta=\)
In a quadrilateral ABCD, \(\angle A = \frac{2\pi}{3}\) and AC is the bisector of angle A. If \(15|AC| = 5|AD| = 3|AB|\), then the angle between \(\vec{AB}\) and \(\vec{BC}\) is
\(\vec{a}, \vec{b}, \vec{c}\) are three non-coplanar and mutually perpendicular vectors of same magnitude K. \(\vec{r}\) is any vector satisfying \(\vec{a}\times((\vec{r}-\vec{b})\times\vec{a}) + \vec{b}\times((\vec{r}-\vec{c})\times\vec{b}) + \vec{c}\times((\vec{r}-\vec{a})\times\vec{c}) = \vec{0}\), then \(\vec{r} =\)
Consider the following
Assertion (A): The two lines \(\vec{r} = \vec{a}+t(\vec{b})\) and \(\vec{r}=\vec{b}+s(\vec{a})\) intersect each other.
Reason (R): The shortest distance between the lines \(\vec{r}=\vec{p}+t(\vec{q})\) and \(\vec{r}=\vec{c}+s(\vec{d})\) is equal to the length of projection of the vector \((\vec{p}-\vec{c})\) on \((\vec{q}\times\vec{d})\).
The correct answer is
The mean deviation about median of the numbers \(3x, 6x, 9x, ..., 81x\) is 91, then \(|x|=\)
Functions are formed from the set \(A = \{a_1, a_2, a_3\}\) to another set \(B = \{b_1, b_2, b_3, b_4, b_5\}\). If a function is selected at random, the probability that it is a one-one function is
A and B are two events of a random experiment such that \(P(B)=0.4\), \(P(A \cap \bar{B}) = 0.5\), \(P(A \cup B) + P(A|B) = 1.15\), then \(P(A)=\)
There are two boxes each containing 10 balls. In each box, few of them are black balls and rest are white. A ball is drawn at random from one of the boxes and found that it is black. If the probability that the black ball drawn is from the second box is \(\frac{1}{5}\), then number of black balls in the first box is
In a shelf there are three mathematics and two physics books. A student takes a book randomly. If he randomly takes, successively for three times by replacing the book already taken every time, then the mean of the number of mathematics books which is treated as random variable is
In a Poisson distribution, if \(\frac{P(X=5)}{P(X=2)} = \frac{1}{7500}\) and \(\frac{P(X=5)}{P(X=3)} = \frac{1}{500}\), then the mean of the distribution is
A(2,0), B(0,2), C(-2,0) are three points. Let a, b, c be the perpendicular distances from a variable point P on to the lines AB, BC and CA respectively. If a, b, c are in arithmetic progression, then the locus of P is
When the coordinate axes are rotated about the origin through an angle \(\frac{\pi}{4}\) in the positive direction, the equation \(ax^2+2hxy+by^2=c\) is transformed to \(25x'^2+9y'^2=225\), then \((a+2h+b-\sqrt{c})^2=\)
\(y-x=0\) is the equation of a side of a triangle ABC. The orthocentre and circumcentre of the triangle ABC are respectively (5,8) and (2,3). The reflection of orthocentre with respect to any side of the triangle lies on its circumcircle. Then the radius of the circumcircle of the triangle is
Two families of lines are given by \(ax+by+c=0\) and \(4a^2+9b^2-c^2-12ab=0\). Then the line common to both the families is
Two non parallel sides of a rhombus are parallel to the lines \(x+y-1=0\) and \(7x-y-5=0\). If (1,3) is the centre of the rhombus and one of its vertices \(A(\alpha, \beta)\) lies on \(15x-5y=6\), then one of the possible values of \((\alpha+\beta)\) is
If the equations \(3x^2+2hxy-3y^2=0\) and \(3x^2+2hxy-3y^2+2x-4y+c=0\) represent the four sides of a square, then \(\frac{h}{c}= \)
The radius of the circle having three chords along y-axis, the line \(y=x\) and the line \(2x+3y=10\) is
Among the chords of the circle \(x^2+y^2=75\), the number of chords having their midpoints on the line \(x=8\) and having their slopes as integers is
The equation of the circle which touches the circle \(S \equiv x^2+y^2-10x-4y+19=0\) at the point (2,3) internally and having radius equal to half of the radius of the circle S=0 is
If \(P(\frac{7}{5}, \frac{6}{5})\) is the inverse point of \(A(1,2)\) with respect to a circle with centre \(C(2,0)\), then the radius of that circle is
If the circle \(S=0\) intersect the three circles \(S_1 = x^2+y^2+4x-7=0\), \(S_2 = x^2+y^2+y=0\) and \(S_3 = x^2+y^2+\frac{3}{2}x+\frac{5}{2}y-\frac{9}{2}=0\) orthogonally, then the radical axis of \(S=0\) and \(S_1=0\) is
If a tangent to the circle \(x^2+y^2+2x+2y+1=0\) is radical axis of the circles \(x^2+y^2+2gx+2fy+c=0\) and \(2x^2+2y^2+3x+8y+2c=0\), then
If the angle between the tangents drawn to the parabola \(y^2=4x\) from the points on the line \(4x-y=0\) is \(\frac{\pi}{3}\), then the sum of the abscissae of all such points is
The normal at a point on the parabola \(y^2=4x\) passes through a point P. Two more normals to this parabola also pass through P. If the centroid of the triangle formed by the feet of these three normals is G(2,0), then the abscissa of P is
The circumcenter of the equilateral triangle having the three points \(\theta_1, \theta_2, \theta_3\) lying on the ellipse \(\frac{x^2}{a^2}+\frac{y^2}{b^2}=1\) as its vertices is \((r,s)\). Then the average of \(\cos(\theta_1-\theta_2), \cos(\theta_2-\theta_3)\) and \(\cos(\theta_3-\theta_1)\) is
The ellipse \(\frac{x^2}{a^2}+\frac{y^2}{b^2}=1\) (\(b>a\)) is an ellipse with eccentricity \(\frac{1}{\sqrt{2}}\). If the angle of intersection between the ellipse and parabola \(y^2=4ax\) is \(\theta\), then the coordinates of the point \(\frac{20}{3}\) on the ellipse is
The number of common tangents that can be drawn to the curves \(\frac{x^2}{16}-\frac{y^2}{9}=1\) and \(x^2+y^2=16\) is
Let A(\(\alpha\),4,7) and B(3,\(\beta\),8) be two points in space. If YZ plane and ZX plane respectively divide the line segment joining the points A and B in the ratio 2:3 and 4:5, then the point C which divides AB in the ratio \(\alpha:\beta\) externally is
The direction ratios of the line bisecting the angle between the x-axis and the line having direction ratios (3, -1, 5) are
If the plane \(-4x-2y+2z+\alpha=0\) is at a distance of two units from the plane \(2x+y-z+1=0\), then the product of all the possible values of \(\alpha\) is
\(\lim_{x \to 0} \frac{\sqrt[3]{\cos x} - \sqrt{\cos x}}{\sin^2 x} =\)
Let \(f:[-1,2] \to \mathbb{R}\) be defined by \(f(x) = [x^2-3]\) where \([.]\) denotes greatest integer function, then the number of points of discontinuity for the function \(f\) in \((-1,2)\) is
If \(f(x) = \begin{cases} x^2 \cos(\frac{\pi}{x}), & x \neq 0
0, & x=0 \end{cases}\), then at \(x=0\), \(f(x)\) is
(Note: The question image says 'at \(x=2\)', but this is a standard problem about the point \(x=0\). At \(x=2\), the function is clearly differentiable by standard rules. We assume the question meant \(x=0\).)
The set of all values of x for which \(f(x) = ||x|-1|\) is differentiable is
If \(y=f(x)^{g(x)}\) and \(\frac{dy}{dx} = y[H(x)f'(x)+G(x)g'(x)]\), then \(\int \frac{G(x)H(x)f'(x)}{g(x)}dx =\)
If \(x=t-\sin t, y=1-\cos t\) and \(\frac{d^2y}{dx^2}=-1\) at \(t=K, K>0\), then \(\lim_{t \to K} \frac{y}{x} =\)
For the curve \((\frac{x}{a})^n + (\frac{y}{b})^n = 2\), (\(n \in N\) \& \(n>1\)) the line \(\frac{x}{a}+\frac{y}{b}=2\) is
The height of a cone with semi vertical angle \(\pi/3\) is increasing at the rate of 2 units/min. The rate at which the radius of the cone is to be decreased so as to have a fixed volume always is
The function \(f(x)=2x^3-9ax^2+12a^2x+1\) where \(a>0\) attains its local maximum and local minimum at p and q respectively. If \(p^2=q\) then a =
Consider all functions given in List-I in the interval [1,3]. The List-2 has the values of 'c' obtained by applying Lagrange's mean value theorem on the functions of List-1. Match the functions and values of 'c'.

If the percentage error in the radius of a circle is 3, then the percentage error in its area is
If \(I_1 = \int \frac{e^x}{e^{4x}+e^{2x}+1}dx\), \(I_2 = \int \frac{e^{-x}}{e^{-4x}+e^{-2x}+1}dx\), then \(I_2-I_1=\)
If \(\int \frac{\sqrt{1-\sqrt{x}}}{\sqrt{x(1+\sqrt{x})}}dx = 2f(x)-2\sin^{-1}\sqrt{x}+c\), then \(f(x)=\)
If \(\int \frac{3x+2}{4x^2+4x+5}dx = A\log(4x^2+4x+5)+B\tan^{-1}(\frac{2x+1}{2})+c\), then \(A+B=\)
Consider the following
Assertion (A): \(\int \sqrt{x-3}(\sin^{-1}(\log x) + \cos^{-1}(\log x))dx = \frac{\pi}{3}(x-3)^{3/2}+c\)
Reason (R): \(\sin^{-1}(f(x))+\cos^{-1}(f(x))=\frac{\pi}{2}\), \(|f(x)|\le 1\)
The correct answer is
\(\lim_{n \to \infty} \frac{(2n(2n-1)...(n+2)(n+1))^{1/n}}{n} =\)
The area of the region bounded by \(y=x^3\), x-axis, \(x=-2\) and \(x=4\) is
If \(\int_0^{\pi/2} \tan^{14}(x/2)dx = 2\left[\sum_{n=1}^7 f(n) - \frac{\pi}{4}\right]\), then \(f(n)=\)
The differential equation of the family of all circles of radius 'a' is
If the general solution of \((1+y^2)dx = (\tan^{-1}y - x)dy\) is \(x = f(y)+ce^{-\tan^{-1}y}\), then \(f(y)=\)
The force of mutual attraction between any two objects by virtue of their masses is
The error in the measurement of force acting normally on a square plate is 3%. If the error in the measurement of the side of the plate is 1%, then the error in the determination of the pressure acting on the plate is
For a particle moving along a straight line path, the displacements in third and fifth seconds of its motion are 10 m and 18 m respectively. The speed of the particle at time t=4s is
The vertical displacement (y in metre) of a projectile in terms of its horizontal displacement (x in metre) is given by \(y=(\sqrt{3}x - 0.2x^2)\). The time of flight of the projectile is (Acceleration due to gravity = 10 ms\(^{-2}\))
A block of mass \(\sqrt{2}\) kg is placed on a rough horizontal surface. A force 'F' acting upwards at an angle of 45\(^\circ\) with the horizontal causes the block to start motion. If the coefficient of static friction between the surface and the block is 0.25, the magnitude of the force 'F' is (Acceleration due to gravity = 10 ms\(^{-2}\))
If the kinetic energy of a body moving with a velocity of \((2\vec{i}+3\vec{j}-4\vec{k})\) ms\(^{-1}\) is 87 J, then the mass of the body is
A body of mass 0.5 kg is supplied with a power 'P' (in watt) which varies with time 't' (in second) as \(P=3t^2+3\). If the velocity of the body at time t=0 is zero, then the velocity of the body at time t=3s is
A solid sphere of mass 2 kg and radius 0.5 m is rolling without slipping on a horizontal surface. The ratio of the rotational and translational kinetic energies of the sphere is
If the length of a thin uniform rod is 'L' and the radius of gyration of the rod about an axis perpendicular to its length and passing through one end is K, then K:L=
The force (F in newton) acting on a particle of mass 90 g executing simple harmonic motion is given by \(F+0.04\pi^2y=0\), where y is displacement of the particle in meter. If the amplitude of the particle is \(\frac{6}{\pi}\) m, then the maximum velocity of the particle is
Which of the following is incorrect about the gravitational force between two bodies?
A steel rod with a circular cross-section of diameter 1cm and another steel rod with a square cross-section of side 1cm have equal mass. If the two rods are subjected to same tension, the ratio of the elongations of the two rods is
A cube of side 40 cm is floating with \(\frac{1}{4}\)th of its volume immersed in water. When a circular disc is placed on the cube, it floats with \(\frac{2}{5}\)th of its volume immersed in water. The mass of the disc is
The maximum length of water column that can stay without falling in a vertically held capillary tube of diameter 1mm and open at both the ends is (Acceleration due to gravity = 10 ms\(^{-2}\) and surface tension of water = 0.07 Nm\(^{-1}\))
A steel pendulum clock manufactured at 32\(^\circ\)C and working at 47\(^\circ\)C is nearly (Coefficient of linear expansion of steel = \(12 \times 10^{-6}\) /\(^\circ\)C)
A metal metre scale that is accurate up to 0.5 mm is made at a temperature of 25\(^\circ\)C. The range of temperatures within which it can be used is (Coefficient of linear expansion of the metal = \(10^{-5}\) /\(^\circ\)C)
A Carnot engine uses diatomic gas as a working substance. During the adiabatic expansion part of the cycle, if the volume of the gas becomes 32 times its initial volume, then the efficiency of the engine is
The ratio of the average translational kinetic energies of hydrogen and oxygen at the same temperature is
The air columns in two tubes closed at one end vibrating in their fundamental modes produce 2 beats per second. The number of beats produced per second when the same tubes are vibrated in their fundamental mode with their both ends open are
A car moving towards a cliff emits sound of frequency 'n'. If the difference in frequencies of the horn and its echo heard by the driver of the car is 10% of 'n', then the speed of the car is nearly (Speed of sound in air is 336ms\(^{-1}\))
A straight metal rod of length 6 cm is placed along the principal axis of a concave mirror of focal length 9 cm such that the end of the rod closer to the mirror is at a distance of 15 cm from the pole of the mirror. The length of the image of the rod is
A ray of light incidents at an angle of \(9.3^\circ\) on one face of a small angle prism of refracting angle \(6^\circ\). If the ray of light emerges normally from the second face, the refractive index of the material of the prism is
The distance for which ray optics becomes a good approximation for an aperture of 0.3 cm and a light of wavelength 6000 \(\AA\) is
The electrostatic force between two charges kept in air is F. If 30% of the space between the charges is filled with a medium, then the electrostatic force between the charges becomes \(\frac{F}{2.56}\). The dielectric constant of the medium is
729 small identical spheres each charged to an electric potential 3V combine to form a bigger sphere. The electric potential of the bigger sphere is
The current amplification factor of a transistor in common emitter configuration is 80. If the emitter current is 2.43 mA, then the base current is

The area of cross-section of a potentiometer wire is \(6 \times 10^{-7}\) m\(^2\). The potential difference per unit length of the potentiometer wire when it is connected to a cell of negligible internal resistance and a resistor in series is \(0.15\) Vm\(^{-1}\). If the current through potentiometer wire is \(0.3\)A, then the resistivity of the material of the potentiometer wire is

As shown in the figure, a uniform straight wire of length \(30\sqrt{3}\) cm is bent in the form of an equilateral triangle ABC. A uniform magnetic field 2T is applied parallel to the side BC. If the current through the wire is 2A, the magnitude of the force on the side AC is
A proton moving with a velocity of \(8\times 10^5\) ms\(^{-1}\) enters a uniform magnetic field normal to the direction of the magnetic field. If the radius of the circular path of the proton in the magnetic field is \(8.3\) cm, then the magnitude of the magnetic field is (Charge of proton = \(1.6\times 10^{-19}\) C and mass of the proton = \(1.66\times 10^{-27}\) kg)
At a certain place in the magnetic meridian, the earth's magnetic field is twice its vertical component. The ratio of horizontal component of earth's magnetic field and the total magnetic field of the earth at that place is
A coil of resistance \(16 \Omega\) is placed with its plane perpendicular to a uniform magnetic field whose flux (\(\phi\) in \(10^{-3}\) weber) changes with time (t in second) as \(\phi = 5t^2+4t+2\). The induced current at time \(t=6\) seconds is
The small energy losses in transformers due to eddy currents can be reduced by
If the electric field of a plane electromagnetic wave is \(E_z = 60\sin(0.5\times 10^3x + 1.5\times 10^{11}t)\) Vm\(^{-1}\), then the magnetic field of the wave is
In a photoelectric experiment, the slope of the graph drawn between stopping potential along y-axis and frequency of incident radiation along x-axis is (Planck's constant = \(6.6\times 10^{-34}\) Js)
The maximum wavelength of incident radiation required to ionize a hydrogen atom in its ground state is nearly
When an element \(_{90}^{232}Th\) decays into \(_{82}^{208}Pb\), the number of \(\alpha\) and \(\beta^{-}\) particles emitted respectively are
During the disintegration of a radioactive nucleus of mass number 208 at rest, two alpha particles each with kinetic energy E are emitted. The total kinetic energy of the emitted alpha particles and the daughter nucleus after the disintegration is
The current amplification factor of a transistor in common emitter configuration is 80. If the emitter current is 2.43 mA, then the base current is
The negative feedback in an amplifier
If the frequencies of the carrier wave and message signal are \(1\) MHz and \(28\) kHz respectively, then the frequencies of the side bands are
Observe the following statements
Statement-I: Rutherford model of an atom cannot explain the stability of an atom
Statement-II: The wavelength of X-rays is higher than the wavelength of microwaves
The correct answer is
In hydrogen atom, an electron is transferred from an orbit of radius \(1.3225\) nm to another orbit of radius \(0.2116\) nm. What is the energy (in J) of emitted radiation? (Rydberg constant \(R_H \approx 1.097 \times 10^7 m^{-1}\))
Identify the correct orders regarding atomic radii
i. \(Cl > F > Li\)
ii. \(P > C > N\)
iii. \(Tm > Sm > Eu\)
iv. \(Sr > Ca > Mg\)
Match the following

The atomic numbers of the elements X, Y, Z are \(a, a+1, a+2\) respectively. Z is an alkali metal. The nature of bonding in the compound formed by X and Z is
The sets of molecules in which central atom has no lone pair of electrons are:
i. \(SnCl_2, NH_3, SF_4\)
ii. \(HgCl_2, SO_3, SF_6\)
iii. \(BeCl_2, BF_3, PCl_5\)
iv. \(ClF_3, BrF_5, XeF_6\)
The isobars of one mole of an ideal gas were obtained at three different pressures \((p_1, p_2\) and \(p_3)\). The slopes of these isobars are \(m_1, m_2\) and \(m_3\) respectively. If \(p_1 < p_2 < p_3\), then the correct relation of the slopes is
\(100 mL\) of \(0.05 M Cu^{2+}\) aqueous solution is added to \(1 L\) of \(0.1 M KI\) solution. The number of moles of \(I_2\) and \(Cu_2I_2\) formed are respectively
The \(C_p\) of an ideal gas is \(10.314 J mol^{-1} K^{-1}\). One mole of this gas is expanded against a constant pressure of \(p atm\). The change in temperature during expansion is \(1.0 K\). The values of \(q\) (in J) and \(\Delta H\) (in \(J mol^{-1}\)) are respectively
At T(K), \(K_p\) value for the reaction, \(2AO_2(g)+O_2(g) \rightleftharpoons 2AO_3(g)\) is \(4\times 10^{10}\). What is the \(K'_p\) value for \(3AO_2(g)+\frac{3}{2}O_2(g) \rightleftharpoons 3AO_3(g)\) at T(K)?
A sample of water contains \(Mg(HCO_3)_2\) and \(Ca(HCO_3)_2\). On boiling this water, these hydrogen carbonates are removed as precipitates. The precipitates are
Which of the following statements is not correct?
The order of negative standard potential values of \(Li, Na, K\) is
In which of the following reactions, hydrogen is evolved?
I. \(Reaction of sodium borohydride with iodine\)
II. \(Oxidation of diborane\)
III. \(Reaction of boron trifluoride with sodium hydride\)
IV. \(Hydrolysis of diborane\)
Which of the following statements is not correct regarding the gas evolved by the reaction of dilute \(HCl\) on \(CaCO_3\)?
Observe the following statements
Statement-I: The carbon containing components of photochemical smog are acrolein, methanal and PAN
Statement-II: The number of greenhouse gases in the list given below is 5
\(CH_4, CO_2, NO, H_2O(l), H_2O(g), O_2, O_3\)
The correct answer is
The condensed, bond line and complete formulae of n-butane are respectively

'X' is the isomer of \(C_6H_{14}\). It has four primary carbons and two tertiary carbons. 'X' can be prepared from which of the following reaction?

What are X and Y respectively in the following reaction sequence?
\[ Iso-pentane \xrightarrow{KMnO_4} X \xrightarrow{\substack{20%\,H_3PO_4
358\,K}} Y \]

What are X and Y respectively in the following reaction sequence?
A metal (M), crystallizes in \(fcc\) lattice with edge length of \(4.242 A\). What is the radius of \(M\) atom (in \(A\))?
A solid mixture weighing \(5 g\) contains equal number of moles of \(Na_2CO_3\) and \(NaHCO_3\). This solid mixture was dissolved in \(1 L\) of water. What is the volume (in \(mL\)) of \(0.1 M HCl\) required to completely react with this \(1 L\) mixture solution?
At \(298 K\) the equilibrium constant for the reaction, \(M(s)+2Ag^+(aq) \rightleftharpoons M^{2+}(aq)+2Ag(s)\) is \(10^{15}\). What is the \(E_{cell}^{\circ}\) (in \(V\)) for this reaction? (\(\frac{2.303 RT}{F} = 0.06 V\))
\(A\to P\) is a first order reaction. At \(300 K\) this reaction was started with \([A]=0.5 molL^{-1}\). The rate constant of reaction was \(0.125 min^{-1}\). The same reaction was started separately with \([A]=1 molL^{-1}\) at \(300 K\). The rate constant (in \(min^{-1}\)) now is
Observe the following reactions
I. \(CO(g)+H_2(g) \xrightarrow{X} HCHO(g)\)
II. \(CO(g)+3H_2(g) \xrightarrow{Y} CH_4(g)+H_2O(g)\)
The catalysts \(X\) and \(Y\) in the above reactions are respectively
Composition of siderite ore is
Which of the following gives more number of oxides on reacting with \(HCl\)?
The number of lone pairs of electrons on the central atom of \(XeO_3, XeOF_4\) and \(XeF_6\) respectively is
Which of the following statements is not correct?
The pair of ions with paramagnetic nature and same number of electrons is
Observe the following complex ions Identify the option in which the unpaired electrons in the complex ions are in correct increasing order

The polymer chains are held together by hydrogen bonding in a polymer X. Polymer X is formed from monomers Y and Z. What are Y and Z?
![]()
Amino acid 'X' contains phenolic hydroxy group and amino acid 'Y' contains amide group. 'X' and 'Y' respectively are
The chemical X is used in the prevention of heart attack. The structure of X is


Observe the following reactions reaction is

Consider the following set of reactions.
\(C_6H_5CN \xrightarrow{B} Y \xrightarrow{A} X (reacts with 2,4-DNP)\)
\(C_6H_5CN \xrightarrow{A} X (dissolves in dil. HCl)\)
What are A and B respectively?

An alcohol X (\(C_5H_{12}O\)) produces turbidity instantly with conc. \(HCl/ZnCl_2\). Isomer (Y) of \(X\) undergoes dehydration with conc. \(H_2SO_4\) at \(443 K\). \(X\) and \(Y\) respectively are
What are \(X\) and \(Y\) in the following reaction sequence?
\(C_5H_{12}O \xrightarrow{Cu/573 K} C_5H_{10} \xrightarrow{(i) O_3, (ii) Zn+H_2O} X + Y\)

Observe the following set of reactions
{l} \(C_6H_5COCl \xrightarrow{X} C_6H_5CHO\)
\(C_6H_5COCl \xrightarrow{Y} C_6H_5COCH_3\)
\(C_6H_5CHO \xrightarrow{OH^-/293 K} Z (Major product)\)
What are \(X, Y\) and \(Z\) respectively?
View Solution
Step 1: Identify reagent X (Reduction of acyl chloride to aldehyde).
The reaction is: \[ C_6H_5COCl \xrightarrow{X} C_6H_5CHO \]
This is the partial reduction of an acyl chloride to an aldehyde.
Reagent \(X\) must be \(H_2/Pd-BaSO_4\) (Rosenmund reduction).
Hence, \(\boxed{X = H_2/Pd-BaSO_4}\)
Step 2: Identify reagent Y (Formation of ketone).
The reaction: \[ C_6H_5COCl \xrightarrow{Y} C_6H_5COCH_3 \]
This can be done using a dialkyl cadmium reagent \((CH_3)_2Cd\) or a Grignard reagent \((CH_3MgBr)\).
Though organocadmium is more specific, the question provides \(CH_3MgBr\) as the option, so we take: \[ \boxed{Y = CH_3MgBr} \]
Step 3: Identify product Z.
The reaction is: \[ C_6H_5CHO \xrightarrow{OH^-/293\,K} Z \]
Under basic conditions, Benzaldehyde can undergo condensation reactions. If the product is given as \(C_6H_5CH=CH-C_6H_5\) (Stilbene), it indicates an aldol-type or Wittig-type condensation product.
Hence, \(\boxed{Z = C_6H_5CH=CH-C_6H_5}\) \; (Stilbene).
Step 4: Final Answer Summary.
\[ \text{X = H_2/Pd-BaSO_4, \quad Y = CH_3MgBr, \quad Z = C_6H_5CH=CH-C_6H_5 \] Quick Tip: The Rosenmund reduction (\(H_2/Pd-BaSO_4\)) is a name reaction for the partial reduction of acyl chlorides to aldehydes. Organometallic reagents like \(R_2Cd\) are best for ketone synthesis. Aldol condensation of aromatic aldehydes with base (like \(C_6H_5CHO\)) is typically a Cannizzaro reaction, unless a strong nucleophile is added.
Consider the following set of reactions.
\(C_6H_5CN \xrightarrow{B} Y (reacts with 2,4-DNP) \xrightarrow{A} X (dissolves in dil. HCl)\)
What are A and B respectively?













































TS EAMCET 2025 Expected Difficulty Level
Based on the analysis of past shifts and expert opinions, the TS EAMCET 2025 Question Paper for the Engineering exam, May 4th 2025 Shift 2, is expected to be of a moderate difficulty level.
| Subject | Expected Difficulty Level | Key Observations |
| Mathematics | Moderate to Tough | It is expected to have Lengthy calculation-based questions with emphasis on vectors, calculus, and coordinate geometry |
| Physics | Moderate | It is expected to have Conceptually based numericals with a focus on thermodynamics, optics, and electrodynamics |
| Chemistry | Easy to Moderate | It will be mostly NCERT-based, with questions from chemical bonding and organic chemistry |
| Overall | Moderate | Balanced paper with a mix of direct and application-based questions |
TS EAMCET 2025 Topic-Wise Weightage
The TS EAMCET 2025 Engineering Exam has a total of 160 multiple-choice questions that are allocated in the following pattern: Mathematics (80), Physics (40), and Chemistry (40).
Understanding of topic-wise weightage helps candidates to estimate the topics that can be asked in the TS EAMCET Question Paper.
Mathematics (80 Questions)
| Topic | Expected Questions | Approximate Weightage (%) |
|---|---|---|
| Algebra | 10–12 | 12% |
| Calculus | 12–14 | 15% |
| Coordinate Geometry | 10–12 | 12% |
| Trigonometry | 8–10 | 10% |
| Probability & Statistics | 8–10 | 10% |
| Vector Algebra | 6–8 | 8% |
Physics (40 Questions)
| Topic | Expected Questions | Approximate Weightage (%) |
|---|---|---|
| Mechanics | 10–12 | 25% |
| Thermodynamics | 6–8 | 15% |
| Electromagnetism | 6–8 | 15% |
| Optics | 4–6 | 10% |
| Modern Physics | 4–6 | 10% |
Chemistry (40 Questions)
| Topic | Expected Questions | Approximate Weightage (%) |
| Organic Chemistry | 12–14 | 30% |
| Inorganic Chemistry | 10–12 | 25% |
| Physical Chemistry | 12–14 | 30% |








Comments