The TS EAMCET 2025 Engineering exam was conducted on May 2, 2025, from 3:00 P.M. to 6:00 P.M. The overall difficulty level of the paper was moderate to tough.
The TS EAMCET Question Paper with Solution PDF is available here for download.
As per the exam pattern, the TS EAMCET 2025 exam was conducted in CBT mode with 160 MCQs in a total of 160 marks in 3 hours (180 minutes), 1 mark is awarded for each correct answer, and no negative marking for incorrect answers.
TS EAMCET 2025 Question Paper with Solution PDF
| TS EAMCET 2025 May 2 Shift 2 Question Paper with Answer Key | Download PDF | Check Solution |

If \( f: \mathbb{R}-\{0\} \to \mathbb{R} \) is defined by \( 3f(x) + 4f\left(\frac{1}{x}\right) = \frac{2-x}{x} \), then \( f(3) = \)
The inverse of the function \( y = \frac{10^x - 10^{-x}}{10^x + 10^{-x}} + 1 \) is \( x = \)
The value of the greatest positive integer \( k \), such that \( 49^k + 1 \) is a factor of \( 48(49^{125} + 49^{124} + \dots + 49^2 + 49 + 1) \) is
If \( \left| \begin{matrix} 1 & 2 & 3-\lambda
0 & -1-\lambda & 2
1-\lambda & 1 & 3 \end{matrix} \right| = A\lambda^3 + B\lambda^2 + C\lambda + D \), then \( D+A = \)
If \( A+2B = \begin{bmatrix} 1 & 2 & 0
6 & -3 & 3
-5 & 3 & 1 \end{bmatrix} \) and \( 2A-B = \begin{bmatrix} 2 & -1 & 5
2 & -1 & 6
0 & 1 & 2 \end{bmatrix} \), then \( tr(A) - tr(B) = \)
A, C are \( 3 \times 3 \) matrices. B, D are \( 3 \times 1 \) matrices. If \( AX=B \) has a unique solution and \( CX=D \) has an infinite number of solutions, then
A and B are two non-square matrices. If \( P = A + B \), \( Q = A^TB \), \( R = AB^T \), then the matrices whose order is equal to the order of A are
\( \omega \) is a complex cube root of unity and \( Z \) is a complex number satisfying \( |Z-1| \le 2 \). The possible values of \( r \) such that \( |Z-1| \le 2 \) and \( |\omega Z - 1 - \omega^2| = r \) have no common solution are
If \( |Z|=2 \), \( Z_1 = \frac{Z}{2}e^{i\alpha} \) and \( \theta \) is the amp(Z), then \( \frac{Z_1^n - Z_1^{-n}}{Z_1^n + Z_1^{-n}} = \)
If \( n, K \in \mathbb{N} \) such that \( n \neq 3K \), then \( (\sqrt{3}+i)^{2n} + (\sqrt{3}-i)^{2n} = \)
In Argand plane, no value of \( \sqrt[3]{1-i\sqrt{3}} \) lie in
If \( l \) is the maximum value of \( -3x^2+4x+1 \) and \( m \) is the minimum value of \( 3x^2+4x+1 \), then the equation of the hyperbola having foci at \( (l,0), (7m,0) \) and eccentricity as 2 is
If the equation \( x^2 - 3ax + a^2 - 2a - K = 0 \) has different real roots for every rational number \( a \), then \( K \) lies in the interval
The number of all common roots of the equation \( x^4 - 10x^3 + 37x^2 - 60x + 36 = 0 \) and the transformed equation of it obtained by increasing any two distinct roots of it by 1, keeping the other two roots fixed, is
If \( \alpha, \beta, \gamma \) are the roots of the equation \( x^3 - Px^2 + Qx - R = 0 \) and \( (\alpha-2)^2, (\beta-2)^2, (\gamma-2)^2 \) are the roots of the equation \( x^3 - 5x^2 + 4x = 0 \), then the possible least value of \( P+Q+R \) is
The number of non negative integral solutions of the equation \( x+y+z+t=10 \) when \( x \ge 2, z \ge 5 \) is
The number of integers lying between 1000 and 10000 such that the sum of all the digits in each of those numbers becomes 30 is
If all the letters of the word MOST are permuted and the words (with or without meaning) thus obtained are arranged in the dictionary order then the rank of the word STOM when counted from the rank of the word MOST, is
The constant term in the expansion of \( \left(1+\frac{1}{x}\right)^{20} \left(30x(1+x)^{29} + (1+x)^{30}\right) \) is
When \( |x| > 3 \), the coefficient of \( \frac{1}{x^n} \) in the expansion of \( x^{3/2} (3+x)^{1/2} \) is
If \( \frac{x^2-3}{(x+2)(x^2+1)} = \frac{A}{x+2} + \frac{Bx+C}{x^2+1} \), then \( 3A+2B-C = \)
If \( 5\sin\theta + 3\cos\left(\theta + \frac{\pi}{3}\right) + 3 \) lies between \( \alpha \) and \( \beta \) (including \( \alpha, \beta \) also), then \( (\alpha-\beta)(\alpha+\beta-6) = \)
\( \frac{\sin 1^\circ + \sin 2^\circ + \dots + \sin 89^\circ}{2(\cos 1^\circ + \cos 2^\circ + \dots + \cos 44^\circ) + 1} = \)
If \( 3\sin(\alpha-\beta) = 5\cos(\alpha+\beta) \) and \( \alpha+\beta \neq \frac{\pi}{2} \), then \( \frac{\tan(\frac{\pi}{4}-\alpha)}{\tan(\frac{\pi}{4}-\beta)} = \)
1 + \cos x + \cos^2 x + \cos^3 x + \dots to \infty = 4+2\sqrt{3}, then x =
Consider the following statements:
Assertion (A): When \( x, y, z \) are positive numbers, then \[ \tan^{-1}\left( \sqrt{\frac{x(x+y+z)}{yz}} \right) + \tan^{-1}\left( \sqrt{\frac{y(x+y+z)}{xz}} \right) + \tan^{-1}\left( \sqrt{\frac{z(x+y+z)}{xy}} \right) = \pi \]
Reason (R): \( \tan^{-1}a + \tan^{-1}b = \tan^{-1}\left( \frac{a+b}{1-ab} \right) \) if \( a > 0 \) and \( b > 0 \).
If \( e^{(\sinh^{-1} 2 + \cosh^{-1} \sqrt{6})} = a + (b+\sqrt{c})\sqrt{a} + b\sqrt{c} \), then \( a+b+c = \)
In a triangle ABC, if \( r_1=4 \), \( r_2=8 \) and \( r_3=24 \), then \( a:b:c = \)
In a triangle ABC, \( (r_2 + r_3)\sec^2\left(\frac{A}{2}\right) = \)
(Note: Based on the answer key and standard identities, the function is interpreted as \( \sec^2 \)).
A, B, C, D are any four points. If E and F are mid points of AC and BD respectively, then \( \vec{AB}+\vec{CB}+\vec{CD}+\vec{AD} = \)
The four points whose position vectors are given by \( 2\bar{a}+3\bar{b}-\bar{c} \), \( \bar{a}-2\bar{b}+3\bar{c} \), \( 3\bar{a}+4\bar{b}-2\bar{c} \) and \( \bar{a}-6\bar{b}+6\bar{c} \) are
If \( a=|\bar{a}| \); \( b=|\bar{b}| \) then \( \left(\frac{\bar{a}}{a^2} - \frac{\bar{b}}{b^2}\right)^2 = \)
\( \bar{a}, \bar{b}, \bar{c} \) are three unit vectors such that \( x\bar{a} + y\bar{b} + z\bar{c} = p(\bar{b} \times \bar{c}) + q(\bar{c} \times \bar{a}) + r(\bar{a} \times \bar{b}) \). If \( (\bar{a},\bar{b})=(\bar{b},\bar{c})=(\bar{c},\bar{a})=\frac{\pi}{3} \), \( (\bar{a}, \bar{b} \times \bar{c})=\frac{\pi}{6} \) and \( \bar{a}, \bar{b}, \bar{c} \) form a right-handed system, then \( \frac{x+y+z}{p+q+r} = \)
Let A be a point having position vector \( \vec{i}-3\vec{j} \) and \( \bar{r} = (\vec{i}-3\vec{j}) + t(\vec{j}-2\vec{k}) \) be a line. If P is a point on this line and is at a minimum distance from the plane \( \bar{r} \cdot (2\vec{i}+3\vec{j}+5\vec{k}) = 0 \), then the equation of the plane through P and perpendicular to AP is
If the variance of the numbers \( 9, 15, 21, \dots, (6n+3) \) is P, then the variance of the first \( n \) even numbers is
Let \( P = \begin{bmatrix} 1 & 2 & 3
4 & 5 & 6
7 & 8 & 9 \end{bmatrix} \) be a matrix. Three elements of this matrix P are selected at random. A is the event of having the three elements whose sum is odd. B is the event of selecting the three elements which are in a row or column. Then \( P(A) + P(A|B) = \)
A, B1, B2, B3 are the events in a random experiment. If \( P(B_1)=0.25, P(B_2)=0.30, P(B_3)=0.45 \), \( P(A|B_1)=0.05, P(A|B_2)=0.04, P(A|B_3)=0.03 \), then \( P(B_2|A) = \)
A, B are the events in a random experiment. If \( P(A)=\frac{1}{2}, P(B)=\frac{1}{3}, P(A \cap B)=\frac{1}{4} \), then \( P(A^c | B^c) + P(A | B) = \)
Two persons A and B play a game by throwing two dice. If the sum of the numbers appeared on the two dice is even, A will get \( \frac{1}{2} \) point and B will get \( \frac{1}{2} \) point. If the sum is odd, A will get one point and B will get no point. The arithmetic mean of the random variable of the number of points of A is
A typist claims that he prepares a typed page with typo errors of 1 per 10 pages. In a typing assignment of 40 pages, if the probability that the typo errors are at most 2 is p, then \(e^2 p =\)
A line segment joining a point A on x-axis to a point B on y-axis is such that AB = 15. If P is a point on AB such that \(\frac{AP}{PB} = \frac{2}{3}\), then the locus of P is
The point \(P(\alpha, \beta)\) (\(\alpha>0, \beta>0\)) undergoes the following transformations successively.
a) Translation to a distance of 3 units in positive direction of x-axis.
b) Reflection about the line \(y=-x\).
c) Rotation of axes through an angle of \(\frac{\pi}{4}\) about the origin in the positive direction.
If the final position of that point P is \((-4\sqrt{2}, -2\sqrt{2})\), then \((\alpha + \beta) =\)
If the line passing through the point \((4, -3)\) and having negative slope makes an angle of \(45^\circ\) with the line joining the points \((1,1), (2,3)\) then the sum of intercepts of that line is
O(0,0), B(-3,-1), C(-1,-3) are vertices of a triangle OBC. D is a point on OC and E is a point on OB. If the equation of DE is \(2x + 2y + \sqrt{2} = 0\), then the ratio in which the line DE divides the altitude of the triangle OBC is
Every point on the curve \(3x + 2y - 3xy = 0\) is the centroid of a triangle formed by the coordinate axes and a line (L) intersecting both the coordinate axes. Then all such lines (L)
The value of 'a' for which the equation \((a^2-3)x^2 + 16xy - 2ay^2 + 4x - 8y - 2 = 0\) represents a pair of perpendicular lines is
The slope of a common tangent to the circles \(x^2 + y^2 = 16\) and \((x-9)^2 + y^2 = 16\) is
The equation of the circle whose radius is 3 and which touches the circle \(x^2 + y^2 - 4x - 6y - 12 = 0\) internally at \((-1, -1)\) is
Suppose C1 and C2 are two circles having no common points, then
The locus of the centre of the circle touching the x-axis and passing through the point \((-1,1)\) is
The centres of all circles passing through the points of intersection of the circles \(x^2 + y^2 + 2x - 2y + 1 = 0\) and \(x^2 + y^2 - 2x + 2y - 2 = 0\) and having radius \(\sqrt{14}\) lie on the curve
A circle S given by \(x^2 + y^2 - 14x + 6y + 33 = 0\) cuts the X-axis at A and B (OB \(>\) OA). C is midpoint of AB. L is a line through C and having slope \((-1)\). If L is the diameter of a circle S' and also the radical axis of the circles S and S', then the equation of the circle S' is
For the parabola \(y = x^2 - 3x + 2\), match the items in list-1 to that of the items in list-2.
S is a focus, Z is intersection of axis and directrix, P is one end point of latus rectum, Q is the point on the parabola at which tangent is parallel to X-axis
List-1 \hspace{4cm List-2
A \hspace{0.5cm P \hspace{3.5cm I \hspace{0.5cm \((2,0)\)
B \hspace{0.5cm Q \hspace{3.5cm II \hspace{0.4cm \((\frac{3}{2}, -\frac{1}{4})\)
C \hspace{0.5cm S \hspace{3.5cm III \hspace{0.3cm \((\frac{3}{2}, 0)\)
D \hspace{0.5cm Z \hspace{3.5cm IV \hspace{0.3cm \((\frac{3}{2}, -\frac{1}{2})\)
\hspace{5cm V \hspace{0.4cm \((0, \frac{3}{2})\)
The locus of a point which divides the line segment joining the focus and any point on the parabola \(y^2 = 12x\) in the ratio \(m:n\) (\(m+n \ne 0\)) is a parabola. Then the length of the latus rectum of that parabola is
The curve represented by \(\frac{x^2}{12-\alpha} + \frac{y^2}{\alpha-10} = 1\) is
If any tangent drawn to the ellipse \(\frac{x^2}{16} + \frac{y^2}{9} = 1\) touches one of the circles \(x^2 + y^2 = \alpha^2\), then the range of \(\alpha\) is
Let \(x\) be the eccentricity of a hyperbola whose transverse axis is twice its conjugate axis. Let \(y\) be the eccentricity of another hyperbola for which the distance between the foci is 3 times the distance between its directrices. Then \(y^2 - x^2 =\)
O(0,0,0), A(3,1,4), B(1,3,2) and C(0,4,-2) are the vertices of a tetrahedron. If G is the centroid of the tetrahedron and \(G_1\) is the centroid of its face ABC, then the point which divides \(GG_1\) in the ratio 1:2 is
If L is a line common to the planes \(3x + 4y + 7z = 1\), \(x - y + z = 5\) then the direction ratios of the line L are
If the points \((1, 1, \lambda)\) and \((-3, 0, 1)\) are equidistant from the plane \(3x + 4y - 12z + 13 = 0\), then the values of \(\lambda\) are
If \(f(x) = \frac{x(a^x - 1)}{1 - \cos x}\) and \(g(x) = \frac{x(1 - a^x)}{a^x \left(\sqrt{1 - x^2} - \sqrt{1 + x^2}\right)}\), then \(\lim_{x \to 0} (f(x) - g(x)) =\)
If \(f(x) = \begin{cases} \frac{a\sin x - bx + cx^2 + x^3}{2\log(1+x) - 2x^3 + x^4} & , x \neq 0
0 & , x = 0 \end{cases}\) is continuous at \(x = 0\), then
If the function \(g(x) = \begin{cases} K\sqrt{x+1} & , 0 \le x \le 3
mx + 2 & , 3 < x \le 5 \end{cases}\) is differentiable, then \(K + m =\)
Consider the following statements
Assertion (A): For \(x \in \mathbb{R} - \{1\}\), \(\frac{d}{dx}\left(\tan^{-1}\left(\frac{1+x}{1-x}\right)\right) = \frac{d}{dx}(\tan^{-1}x)\)
Reason (R): For \(x < 1\), \(\tan^{-1}\left(\frac{1+x}{1-x}\right) = \frac{\pi}{4} + \tan^{-1}x\),
for \(x > 1\), \(\tan^{-1}\left(\frac{1+x}{1-x}\right) = -\frac{3\pi}{4} + \tan^{-1}x\)
The correct answer is
If \(\frac{d}{dx}\left\{ \frac{x-1}{x-\sqrt{x}} e^{2x+1} \right\} = \frac{x-1}{x-\sqrt{x}} e^{2x+1} f(x)\), then \(f(4) =\)
If \(y = (\sin^{-1}x)^2\), then \((1-x^2)\frac{d^2y}{dx^2} - x\frac{dy}{dx} =\)
The radius of a cone of height 9 units is changed from 2 units to 2.12 units. The exact change and approximate change in the volume of the cone are respectively
The local maximum value \(l\) and local minimum value \(m\) of \(f(x) = \frac{x^2+2x+2}{x+1}\) in \(\mathbb{R} - \{-1\}\) exist at \(\alpha, \beta\) respectively, then \(\frac{l+m}{\alpha+\beta} =\)
\(P(5,2)\) is a point on the curve \(y=f(x)\) and \(\frac{7}{2}\) is the slope of the tangent to the curve at P. The area of the triangle formed by the tangent and the normal to the curve at P with x-axis is
If a particle is moving in a straight line so that after \(t\) seconds its distance \(S\) (in cms) from a fixed point on the line is given by \(S = f(t) = t^3 - 5t^2 + 8t\) then the acceleration of the particle at \(t=5\) sec is (in cm/sec\(^2\))
If \(f:[a,b] \to [c,d]\) is a continuous and strictly increasing function, then \(\frac{d-c}{b-a}\) is
\(\int \left( \frac{1}{x^2} + \frac{\sin^3 x + \cos^3 x}{\sin^2 x \cos^2 x} \right) dx =\)
If \(I_n = \int \frac{1}{(x^2+1)^n} dx\), then \(2n I_{n+1} - (2n-1) I_n =\)
\(\int \frac{x^3}{x^4 + 3x^2 + 2} dx =\)
If \(\int \frac{dx}{(x^2+9)\sqrt{x^2+16}} = \frac{1}{3\sqrt{7}} \tan^{-1} \left( K \frac{x}{\sqrt{16+x^2}} \right) + c\), then \(K =\)
\(\lim_{n \to \infty} \frac{1}{n^2} \left[ e^{1/n} + 2e^{2/n} + 3e^{3/n} + \dots + 2n e^{2n/n} \right] =\)
Let m, n, p, q be four positive integers. If \(\int_0^{2\pi} \sin^m x \cos^n x dx = 4 \int_0^{\pi/2} \sin^m x \cos^n x dx\), \(\int_0^{2\pi} \sin^p x \cos^q x dx = 0\), \(a = m+n+p\) and \(b = m+n+q\), then
The area of the region bounded by the curves \(y=x^3\), \(y=x^2\) and the lines \(x=0\) and \(x=2\) is
The substitution required to reduce the differential equation \(t^2 dx + (x^2 - tx + t^2) dt = 0\) to a differential equation which can be solved by variables separable method is
The equation which represents the system of parabolas whose axis is parallel to y-axis satisfies the differential equation
Bose-Einstein statistics is applicable to particles with
If L and C are inductance and capacitance respectively, then the dimensional formula of \((LC)^{-\frac{1}{2}}\) is
The ratio of times taken by a freely falling body to travel first 5m, second 5m, third 5m distances is
Two bodies are projected from the same point with the same initial velocity 'u' making angles '\(\theta\)' and (\(90^\circ-\theta\)) with the horizontal in opposite directions. The horizontal distance between their positions when the bodies are at their maximum heights is
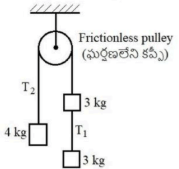
If the system of blocks shown in the figure is released from rest, the ratio of the tensions \(T_1\) and \(T_2\) is (Neglect the mass of the string shown in the figure)
If the component of the vector \(\vec{A}\) along the vector \(\vec{B}\) is twice the component of \(\vec{B}\) along \(\vec{A}\), then the ratio of magnitudes of vectors \(\vec{A}\) and \(\vec{B}\) is
A body projected vertically up with an initial speed of \(10 ms^{-1}\) reaches the point of projection after sometime with a speed of \(8 ms^{-1}\). The maximum height reached by the body is (Acceleration due to gravity \(= 10 ms^{-2}\))
Due to global warming, if the ice in the polar region melts and some of this water flows to the equatorial region, then
If the moment of inertia of a thin circular ring about an axis passing through its edge and perpendicular to its plane is I, then the moment of inertia of the ring about its diameter is
A particle is executing simple harmonic motion. If the force acting on the particle at a position is 86.6% of the maximum force on it, then the ratio of its velocity at that point and its maximum velocity is
The ratio of the time periods of a simple pendulum at heights \(2R_E\) and \(3R_E\) from the surface of the earth is (\(R_E\) is radius of the earth)
Two wires A and B made of same material and areas of cross-section in the ratio \(1:2\) are stretched by same force. If the masses of the wires A and B are in the ratio \(2:3\), then the ratio of the elongations of the wires A and B is
Water is filled in a tank up to a height of 20 cm from the bottom of the tank. Water flows through a hole of area \(1 mm^2\) at its bottom. The mass of the water coming out from the hole in a time of 0.6 s is
(Density of water \(= 1000 kg m^{-3}\) and acceleration due to gravity \(= 10 ms^{-2}\))
For which of the following Reynold's number, a flow is streamlined?
A body cools from a temperature of \(60\,^\circ\mathrm{C}\) to \(50\,^\circ\mathrm{C}\) in 10 minutes and \(50\,^\circ\mathrm{C}\) to \(40\,^\circ\mathrm{C}\) in 15 minutes. The time taken in minutes for the body to cool from \(40\,^\circ\mathrm{C}\) to \(30\,^\circ\mathrm{C}\) is
When the temperature of a gas in a closed vessel is increased by \(2.4\,^\circ\mathrm{C}\), its pressure increases by \(0.5%\). The initial temperature of the gas is
A gas is suddenly compressed such that its absolute temperature is doubled. If the ratio of the specific heat capacities of the gas is 1.5, then the percentage decrease in the volume of the gas is
If the heat required to increase the rms speed of 4 moles of a diatomic gas from \(v\) to \(\sqrt{3}v\) is \(83.1\,\mathrm{kJ}\), then the initial temperature of the gas is (Universal gas constant \(= 8.31\,\mathrm{J\,mol^{-1}\,K^{-1}}\))
If the lengths of the open and closed pipes are in the ratio of \(2:3\), then the ratio of the frequencies of the third harmonic of the open pipe and the fifth harmonic of the closed pipe is
The equation of a transverse wave propagating on a stretched string is given by \(y = 3\sin(4x + 200t)\), where \(x\) and \(y\) are in metre and the time \(t\) is in second. If the tension applied to the string is \(500\,\mathrm{N}\), the linear density of the string is
A compound microscope has an objective of focal length \(1.25\,\mathrm{cm}\) and an eyepiece of focal length \(5\,\mathrm{cm}\) separated by a distance of \(7.5\,\mathrm{cm}\). The total magnification produced by the microscope when the final image forms at infinity is
The property of light that explains the formation of coloured images due to thick lenses is
For an aperture of \(5 \times 10^{-3}\,\mathrm{m}\) and a monochromatic light of wavelength \(\lambda\), the distance for which ray optics becomes a good approximation is \(50\,\mathrm{m}\), then \(\lambda =\)
An electron and a positron enter a uniform electric field E perpendicular to it with equal speeds at the same time. The distance of separation between them in the direction of the field after a time 't' is
(\(\frac{e}{m}\) is specific charge of electron)
A charge q is placed at the centre 'O' of a circle of radius R and two other charges q and q are placed at the ends of the diameter AB of the circle. The work done to move the charge at point B along the circumference of the circle to a point C as shown in the figure is
In a potentiometer experiment, a wire of length 10 m and resistance \(5\,\Omega\) is connected to a cell of emf 2.2 V. If the potential difference between two points separated by a distance of 660 cm on potentiometer wire is 1.1 V, then the internal resistance of the cell is
When the right gap of a meter bridge consists of two equal resistors in series, the balancing point is at 50 cm. When one of the resistors in the right gap is removed and is connected in parallel to the resistor in the left gap, the balancing point is at
Two identical wires, carrying equal currents are bent into circular coils A and B with 2 and 3 turns respectively. The ratio of the magnetic fields at the centres of the coils A and B is
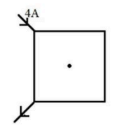
A current of 4 A is passed through a square loop of side 5 cm made of a uniform manganin wire as shown in the figure. The magnetic field at the centre of the loop is
If \(B_V\) and \(B_H\) are respectively the vertical and horizontal components of the earth's magnetic field at a place where the angle of dip is \(60^\circ\), then the total magnetic field at that place is
A coil of resistance \(8\,\Omega\), number of turns 250 and area \(120 cm^2\) is placed in a uniform magnetic field of 2 T such that the plane of the coil makes an angle of \(\frac{\pi}{6}\) with the direction of the magnetic field. In a time of 100 ms, the coil is rotated until its plane becomes parallel to the direction of the magnetic field. The current induced in the coil is
An inductor and a resistor are connected in series to an ac supply. If the potential differences across the inductor and the resistor are 180 V and 240 V respectively, then the voltage of the ac supply is
If electromagnetic waves of power 600 W incident on a non-reflecting surface, then the total force acting on the surface is
When a photosensitive material is illuminated by photons of energy 3.1 eV, the stopping potential of the photoelectrons is 1.7 V. When the same photosensitive material is illuminated by photons of energy 2.5 eV, the stopping potential of the photoelectrons is
The ratio of the kinetic energies of the electrons in the third and fourth excited states of hydrogen atom is
In \(\beta^-\) decay, a neutron transforms into a proton within the nucleus according to the equation:
neutron \(\to\) proton + \(\beta^-\) + \(x\)
In this equation the particle represented by 'x' is
Two radioactive substances A and B have same number of initial nuclei. If the half-lives of A and B are 1.5 days and 4.5 days respectively, then the ratio of the number of nuclei remaining in A and B after 9 days is
\(10^{10}\) electrons enter the emitter of a junction transistor in a time of \(0.4\ \mus\). If \(5%\) of the electrons are lost in the base, then the collector current is
An electron in n-region of a p-n junction moves towards the junction with a speed of \(5 \times 10^5\ ms^{-1}\). If the barrier potential of the junction is \(0.45\ V\), then the speed with which the electron enters the p-region after penetration through the barrier is
(Charge of the electron \(= 1.6 \times 10^{-19}\ C\) and mass of the electron \(= 9 \times 10^{-31}\ kg\))
Coaxial cable, a widely used wire medium offers an approximate frequency bandwidth of
The electron in hydrogen atom undergoes transition from higher orbits to an orbit of radius 476.1 pm. This transition corresponds to which of the following series?
Identify the incorrect statement from the following?
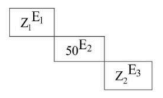
Atomic numbers of three elements \(E_1, E_2\) and \(E_3\) of periodic table are \(Z_1, 50\) and \(Z_2\) respectively. From the position of the elements shown in figure, the value of \((Z_2 - Z_1)\) is
Electron gain enthalpy values (\(\Delta_{eg}H\)) (in kJ mol\(^{-1}\)) of elements X, Y and Z are \(-349, -200\) and \(-295\) respectively. X, Y and Z are respectively
Observe the following list of molecules. Number of polar and non polar molecules are respectively
\(NH_3, BF_3, NF_3, H_2S, CO_2, CH_4, CHCl_3, H_2O\)
The molecule `X' has see-saw shape with central atom in \(sp^3d\) hybridization. What is `X'?
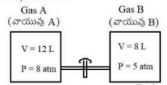
Two vessels are filled with ideal gases A and B and are connected through a pipe of zero volume as shown in figure. The stop cock is opened and the gases are allowed to mix homogeneously and the temperature is kept constant. The partial pressures of A and B respectively (in atm) are
If the number of moles of \(Fe^{2+}\) ions oxidized by one mole of acidified \(MnO_4^-\) is \(x\), the number of moles of \(Fe^{2+}\) ions oxidized by one mole of acidified \(Cr_2O_7^{2-}\) is
One mole of an ideal gas at 300 K and 20 atm expands to 2 atm under isothermal and reversible conditions. The work done by the gas is \(-x\ kJ mol^{-1}\). The value of \(x\) is
\((R=8.3\ J K^{-1}\ mol^{-1})\)
At 1000 K, the equilibrium constant for the reaction
\(CO_2(g) + H_2(g) \rightleftharpoons CO(g) + H_2O(g)\) is 0.53. In a one litre vessel, at equilibrium the mixture contains 0.25 mole of CO, 0.5 mole of \(CO_2\), 0.6 mole of \(H_2\) and \(x\) moles of \(H_2O\). The value of \(x\) is
Match the following
\begin{tabular{|l|l|l|l|
\hline
\multicolumn{2{|c|{List - 1 (Reactions) & \multicolumn{2{c|{List - 2 (Methods)
\hline
A & \(Mg(HCO_3)_2 \rightarrow Mg(OH)_2 \downarrow + 2CO_2 \uparrow\) & I & Clark's method
\hline
B & \(M^{2+} + Na_6P_6O_{18}^{2-} \rightarrow [Na_2MP_6O_{18}]^{2-} + 2Na^+\) & II & Ion exchange method
\hline
C & \(Ca(HCO_3)_2 + Ca(OH)_2 \rightarrow 2CaCO_3 + 2H_2O\) & III & Boiling
\hline
D & \(2NaZ + Ca^{2+}(aq) \rightarrow 2Na^+ + CaZ\) \((Z = Zeolite)\) & IV & Calgon's method
\hline
\end{tabular
The correct answer is
View Solution
Step 1: Understanding the Concept:
The question asks to match reactions involved in removing hardness of water with the specific methods used.
Step 2: Analyzing Each Reaction:
Reaction A: \(Mg(HCO_3)_2 \xrightarrow{\Delta} Mg(OH)_2 \downarrow + 2CO_2 \uparrow\).
This reaction represents the removal of temporary hardness caused by magnesium bicarbonate by Boiling. Note that unlike calcium bicarbonate which forms carbonate, magnesium bicarbonate precipitates as hydroxide because \(Mg(OH)_2\) is less soluble than \(MgCO_3\). (Matches with III).
Reaction B: \(M^{2+} + Na_6P_6O_{18}^{2-} \rightarrow [Na_2MP_6O_{18}]^{2-} + 2Na^+\).
The compound \(Na_6P_6O_{18}\) is commercially known as Calgon (Sodium hexametaphosphate). It complexes with metal ions like \(Ca^{2+}\) and \(Mg^{2+}\) to keep them in solution but inactive. This is Calgon's method. (Matches with IV).
Reaction C: \(Ca(HCO_3)_2 + Ca(OH)_2 \rightarrow 2CaCO_3 \downarrow + 2H_2O\).
This involves adding calculated amounts of lime (\(Ca(OH)_2\)) to precipitate out calcium carbonate. This is known as Clark's method. (Matches with I).
Reaction D: \(2NaZ + Ca^{2+} \rightarrow CaZ_2 + 2Na^+\).
Here, Z represents Zeolite (hydrated sodium aluminium silicate). The exchange of sodium ions for calcium/magnesium ions on the zeolite surface is characteristic of the Ion exchange method (specifically Permutit/Zeolite process). (Matches with II).
Step 3: Matching:
A \(\rightarrow\) III, B \(\rightarrow\) IV, C \(\rightarrow\) I, D \(\rightarrow\) II.
Final Answer: Option (A). Quick Tip: Remember the unique precipitation product for Magnesium temporary hardness on boiling: it forms \(Mg(OH)_2\), not \(MgCO_3\), due to solubility product differences (\(K_{sp}\) of \(Mg(OH)_2 < MgCO_3\)).
Observe the following statements
Statement - I: Both LiF and CsI have low solubility in water
Statement - II: Low solubility of LiF in water is due to smaller hydration enthalpy of ions and that of CsI is due to its high lattice enthalpy
The correct answer is
In which of the following the s-block elements are arranged in the correct order of their melting points?
The correct statements about the compounds of boron are
I. In borax bead test, the colour of cobalt metaborate is blue
II. Diborane is prepared by the oxidation of sodium borohydride with iodine
III. In diborane oxidation state of hydrogen is +1
IV. Boric acid is a tribasic acid
Dehydration of an organic acid X with concentrated \(H_2SO_4\) at 373K gives \(H_2O\) and gas Y. The hybridisation of the carbon in Y and nature of Y are respectively
Identify the correct statements from the following
I. Photochemical smog has high concentration of oxidising agents
II. \(NO_2\) is present in classical smog
III. Higher concentration of \(SO_2\) in air can cause stiffness of flower buds
IV. pH of rain water is approximately 7.5
Consider the given sequence of reactions.
\(C_2H_6 + \frac{3}{2}O_2 \xrightarrow[\Delta]{(CH_3COO)_2Mn} X \xrightarrow{Na} Y\)
Electrolysis of aqueous solution of Y gives gases P and Q at anode. P and Q are respectively.
When sodium fusion extract of an organic compound is boiled with iron (II) sulphate solution followed by addition of concentrated \(H_2SO_4\), gives Prussian blue colour. This confirms the presence of the element
Which of the following is an example of electrophilic substitution reaction?
An alkene X on ozonolysis gives a mixture of simplest ketone (Y) and 3-Pentanone. The IUPAC name of the alkene X is
A solid contains elements A and B. Anions of B form ccp lattice. Cations of A occupy 50% of octahedral voids and 50% of tetrahedral voids. What is the molecular formula of the solid?
The osmotic pressure (in atm) of an aqueous solution containing 0.01 mol of NaCl (degree of dissociation 0.94) and 0.03 mol of glucose in 500 mL at \(27\,^\circC\) is \((R = 0.082\,L atm K^{-1}mol^{-1})\)
Electrolysis of aqueous copper (II) sulphate between Pt electrodes gives 'X' at anode and 'Y' at cathode. X and Y are respectively
Consider a general first order reaction \(A(g) \rightarrow B(g) + C(g)\). If the initial pressure is 200 mm and after 20 minutes it is 250 mm, then the half-life period of the reaction (in minutes) is
(\(\log 2 = 0.30, \log 3 = 0.48, \log 4 = 0.60\))
The most effective coagulating agent for antimony sulphide sol is
Metal X obtained from sphalerite ore can be purified by which of the following methods?
An oxoacid of phosphorus 'X' reduces silver nitrate solution to metallic silver and gets oxidised to another compound Y. X and Y respectively are
Zinc on reaction with concentrated nitric acid gives an oxide of nitrogen (A). Zinc with dilute nitric acid gives another oxide of nitrogen (B). Oxidation numbers of nitrogen in (A) \& (B) are respectively
Identify the reaction related to Deacon's process
Identify the correct statements about lanthanoids
I. \(Ce^{4+}\) and \(Tb^{4+}\) act as oxidising agents
II. \(Eu^{2+}\) and \(Yb^{2+}\) act as oxidising agents
III. Mischmetal is an alloy of 95% iron and 5% lanthanoid metal
IV. \(La^{3+}\) and \(Ce^{4+}\) are diamagnetic in nature
When 100 mL of 0.2 M solution of \(CoCl_3 \cdot xNH_3\) is treated with excess of \(AgNO_3\) solution, \(3.6 \times 10^{22}\) ions are precipitated. The value of \(x\) is \((N = 6 \times 10^{23} mol^{-1})\)
Ethylene on reaction with cold, dilute alkaline \(KMnO_4\) at 273 K gives a compound 'P'. This on polymerisation with which of the following gives dacron?
A carbohydrate (A), when treated with dilute HCl in alcoholic solution gives two isomers (B) and (C). B on reaction with bromine water gives a monocarboxylic acid 'Z' and 'C' is a ketohexose. What is A?
The incorrect statement about chloramphenicol is
The number of chlorine (Cl) atoms in the structure of DDT molecule is
The major product in Reimer-Tiemann reaction is X. The reactants are Y and Z. X, Y and Z are respectively.
Toluene \(\xrightarrow{(1) CrO_2Cl_2 / CS_2} \xrightarrow{(2) H_3O^+} X \xrightarrow[Conc. NaOH]{50%} Y + Z\)
The correct statements about X, Y and Z are
A. Y is a secondary alcohol
B. Y is the reduction product of X
C. Z on heating with soda lime gives benzene
D. Y does not give \(H_2\) gas with Na metal
Identify the product 'P' in the given reaction sequence
![]()
The product 'C' in the given reaction sequence is

The amine / salt of amine which gives positive test with a mixture of chloroform and alcoholic KOH solution is















































































TS EAMCET 2025 Previous Year Exam Analysis
As per the previous years' TS EAMCET Exam Analysis, it is expected that the difficulty level of TS EAMCET 2025 will be moderate in the difficulty level. Below is a year-wise comparison of the TS EAMCET Engineering exams from 2024 to 2022, including subject-wise difficulty, key topics, and student reactions:
TS EAMCET Engineering Exam Analysis (2024–2022)
| Year | Overall Level | Maths Difficulty | Physics Focus | Chemistry Nature |
|---|---|---|---|---|
| 2024 | Moderate | Lengthy, Moderate | Conceptual + Numerical | 65% NCERT-based |
| 2023 | Moderate to Tough | Application-based | 60% Numerical | 70% Direct from NCERT |
| 2022 | Easy to Moderate | Formula-based | 55% Theory-based | 75% Memory-based |
TS EAMCET 2025 Topic-Wise Weightage
TS EAMCET 2025 is expected to follow a question distribution pattern similar to the previous years. Maths has the most weightage at 80 questions, with Physics and Chemistry at 40 each.
Given below is the probable topic-wise weightage of every subject as per the previous year trends:
Mathematics
| Topic | Expected No. of Questions |
|---|---|
| Calculus | 10–12 |
| Algebra | 9–11 |
| Coordinate Geometry | 7–9 |
| Vector Algebra | 5–7 |
| Probability & Statistics | 4–6 |
| Trigonometry | 3–4 |
| Complex Numbers | 2–3 |
Physics
| Topic | Expected No. of Questions |
|---|---|
| Current Electricity | 4–5 |
| Thermodynamics | 4–5 |
| Laws of Motion | 3–4 |
| Work, Energy & Power | 3–4 |
| Semiconductor Devices | 3–4 |
| Oscillations & Waves | 2–3 |
| Modern Physics | 2–3 |
Chemistry
| Topic | Expected No. of Questions |
| Organic Chemistry | 8–10 |
| Physical Chemistry | 7–9 |
| Inorganic Chemistry | 6–8 |
| Environmental Chemistry | 2–3 |
| Surface Chemistry | 2–3 |
| Biomolecules & Polymers | 2–3 |















Comments